Abstract
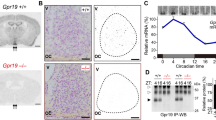
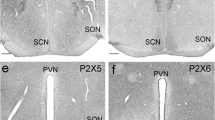
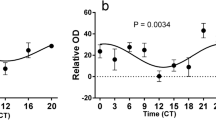
Peroxisome proliferator-activated receptors (PPARs) are members of the nuclear receptor superfamily and function as transcription factors that regulate gene expression in numerous biological processes. Although the PPARβ/δ subtype is highly expressed in the brain, its physiological roles in neuronal function remain to be elucidated. In this study, we examined the presence of PPARβ/δ in the master circadian clock of the Syrian hamster and investigated its putative functional role in this structure. In mammals, the central circadian clock, located in the suprachiasmatic nucleus (SCN), is entrained by the light–dark (LD) cycle via photic6 signals conveyed by a direct pathway whose terminals release glutamate. Using immunocytochemical and qRT-PCR analysis, we demonstrated that the rhythmic expression of PPAR β/δ within the SCN of hamsters raised under an LD cycle was detectable only at the transcriptional level when the hamsters were maintained under constant darkness (DD). The increase in the number of immunoreactive PPARβ/δ cells observed under DD after light stimulation during the early subjective night (CT14), but not during the subjective day (CT06), demonstrated that the expression of PPARβ/δ can be up-regulated according to the photosensitive phase of the circadian clock. All of the PPARβ/δ-positive cells in the SCN also expressed the glutamate receptor NMDAR1. Moreover, we demonstrated that at the photosensitive point (CT14), the administration of L-16504, a specific agonist of PPARβ/δ, amplified the phase delay of the locomotor response induced by a light pulse. Taken together, these data suggest that PPARβ/δ activation modulates glutamate release that mediates entrainment of the circadian clock by light.







Similar content being viewed by others
References
Desvergne B, Wahli W (1999) Peroxisome proliferator-activated receptors: nuclear control of metabolism. Endocr Rev 20(5):649–688
Evans RM, Barish GD, Wang Y-X (2004) PPARs and the complex journey to obesity. Nat Med 10(4):355–361
Yang X, Downes M, Yu RT, Bookout AL, He W, Straume M, Mangelsdorf DJ, Evans RM (2006) Nuclear receptor expression links the circadian clock to metabolism. Cell 126(4):801–810
Lemberger T, Saladin R, Vazquez M, Assimacopoulos F, Staels B, Desvergne B, Wahli W, Auwerx J (1996) Expression of the peroxisome proliferator-activated receptor alpha gene is stimulated by stress and follows a diurnal rhythm. J Biol Chem 271(3):1764–1769
Patel DD, Knight BL, Wiggins D, Humphreys SM, Gibbons GF (2001) Disturbances in the normal regulation of SREBP-sensitive genes in PPARalpha-deficient mice. J Lipid Res 42(3):328–337
Ximenes da Silva A, Lavialle F, Gendrot G, Guesnet P, Alessandri JM, Lavialle M (2002) Glucose transport and utilization are altered in the brain of rats deficient in n-3 polyunsaturated fatty acids. J Neurochem 81(6):1328–1337
Braissant O, Wahli W (1998) Differential expression of peroxisome proliferator-activated receptor-alpha, -beta, and -gamma during rat embryonic development. Endocrinology 139(6):2748–2754
Hall MG, Quignodon L, Desvergne B (2008) Peroxisome proliferator-activated receptor β/δ in the brain: facts and hypothesis. PPAR Res 2008:780452. doi:10.1155/2008/780452
Moreno S, Farioli-Vecchioli S, Cerù MP (2004) Immunolocalization of peroxisome proliferator-activated receptors and retinoid × receptors in the adult rat CNS. Neuroscience 123(1):131–145
Dickmeis T (2009) Glucocorticoids and the circadian clock. J Endocrinol 200(1):3–22
Pevet P, Challet E (2011) Melatonin: both master clock output and internal time-giver in the circadian clocks network. J Physiol Paris 105(4–6):170–182
Reppert SM, Weaver DR (2002) Coordination of circadian timing in mammals. Nature 418(6901):935–941
Ebling FJ (1996) The role of glutamate in the photic regulation of the suprachiasmatic nucleus. Progr Neurobiol 50(2–3):109–132
Bright JJ, Kanakasabai S, Chearwae W, Chakraborty S (2008) PPAR regulation of inflammatory signaling in CNS diseases. PPAR Res 2008:658520. doi:10.1155/2008/658520
Inoue I, Shinoda Y, Ikeda M, Hayashi K, Kanazawa K, Nomura M, Matsunaga T, Xu H, Kawai S, Awata T, Komoda T, Katayama S (2005) CLOCK/BMAL1 is involved in lipid metabolism via transactivation of the peroxisome proliferator-activated receptor (PPAR) response element. J Atheroscler Thromb 12(3):169–174
Canaple L, Rambaud J, Dkhissi-Benyahya O, Ba Rayet, Tan NS, Michalik L, Delaunay F, Wahli W, Laudet V (2006) Reciprocal regulation of brain and muscle Arnt-like protein 1 and peroxisome proliferator-activated receptor alpha defines a novel positive feedback loop in the rodent liver circadian clock. Mol Endocrinol 20(8):1715–1727
Corton JC, Apte U, Anderson SP, Limaye P, Yoon L, Latendresse J, Dunn C, Everitt JI, Voss KA, Swanson C, Kimbrough C, Wong JS, Gill SS, Chandraratna RA, Kwak MK, Kensler TW, Stulnig TM, Steffensen KR, Gustafsson JA, Mehendale HM (2004) Mimetics of caloric restriction include agonists of lipid-activated nuclear receptors. J Biol Chem 279(44):46204–46212
Teboul M, Guillaumond F, Grechez-Cassiau A, Delaunay F (2008) Minireview: the nuclear hormone receptor family round the clock. Mol Endocrinol 22(12):2573–2582
Lavialle M, Begue A, Papillon C, Vilaplana J (2001) Modifications of retinal afferent activity induce changes in astroglial plasticity in the hamster circadian clock. Glia 34:88–100
Shibata S, Watanabe A, Hamada T, Ono M, Watanabe S (1994) N-methyl-d aspartate induces phase shifts in circadian rhythm of neuronal activity of rat SCN in vitro. Am J Physiol 267:R360–R364
Mintz EM, Marvel CL, Gillespie CF, Price KM, Albers HE (1999) Activation of NMDA receptors in the suprachiasmatic nucleus produces light-like phase shifts of the circadian clock in vivo. J Neurosci 19(12):5124–5130
Guan H-P, Ishizuka T, Chui PC, Lehrke M, Lazar MA (2005) Corepressors selectively control the transcriptional activity of PPARgamma in adipocytes. Genes Dev 19(4):453–461
Forman BM, Chen J, Evans RM (1997) Hypolipidemic drugs, polyunsaturated fatty acids, and eicosanoids are ligands for peroxisome proliferator-activated receptors alpha and delta. Proc Natl Acad Sci USA 94(9):4312–4317
Krey G, Braissant O, L’Horset F, Kalkhoven E, Perroud M, Parker MG, Wahli W (1997) Fatty acids, eicosanoids, and hypolipidemic agents identified as ligands of peroxisome proliferator-activated receptors by coactivator-dependent receptor ligand assay. Mol Endocrinol 11(6):779–791
Xu HE, Lambert MH, Montana VG, Parks DJ, Blanchard SG, Brown PJ, Sternbach DD, Lehmann M Jr, Wisely GB, Willson TM, Kliewer SA, Milburn MV (1999) Molecular recognition of fatty acids by peroxisome proliferator-activated receptors. Mol Cell 3(3):397–403
Irwin RP, Allen CN (2007) Calcium response to retinohypothalamic tract synaptic transmission in suprachiasmatic nucleus neurons. J Neurosci 27(43):11748–11757
Yoshihara Y, Watanabe Y (1990) Translocation of phospholipase A2 from cytosol to membranes in rat brain induced by calcium ions. Biochem Biophys Res Commun 170(2):484–490
Lazarewicz JW, Salinska E, Wroblewski JT (1992) NMDA receptor-mediated arachidonic acid release in neurons: role in signal transduction and pathological aspects. Adv Exp Med Biol 318:73–89
Strokin M, Sergeeva M, Reiser G (2003) Docosahexaenoic acid and arachidonic acid release in rat brain astrocytes is mediated by two separate isoforms of phospholipase A2 and is differently regulated by cyclic AMP and Ca2+. Br J Pharmacol 139(5):1014–1022
Basselin M, Chang L, Bell JM, Rapoport SI (2006) Chronic lithium chloride administration attenuates brain NMDA receptor-initiated signaling via arachidonic acid in unanesthetized rats. Neuropsychopharmacology 31(8):1659–1674
Kolko M, Rodriguez de Turco EB, Diemer NH, Bazan NG (2003) Neuronal damage by secretory phospholipase A2: modulation by cytosolic phospholipase A2, platelet-activating factor, and cyclooxygenase-2 in neuronal cells in culture. Neurosci Lett 338(2):164–168
Li SR, Wu KK, Anggard E, Ferns G (1993) Localization of prostaglandin G/H synthase gene expression in rat brain by in situ hybridization. Biol Signals 2(2):77–83
Xu L, Han C, Lim K, Wu T (2006) Cross-talk between peroxisome proliferator-activated receptor delta and cytosolic phospholipase A2alpha/cyclooxygenase-2/prostaglandin E2 signaling pathways in human hepatocellular carcinoma cells. Cancer Res 66(24):11859–11868
Bendová Z, Sumová A, Mikkelsen JD (2009) Circadian and developmental regulation of N-methyl-d-aspartate-receptor 1 mRNA splice variants and N-methyl-d-aspartate-receptor 3 subunit expression within the rat suprachiasmatic nucleus. Neuroscience 159(2):599–609
Colwell CS (2001) NMDA-evoked calcium transients and currents in the suprachiasmatic nucleus: gating by the circadian system. Eur J Neurosci 13:1420–1428
Colwell CS (2000) Circadian modulation of calcium levels in cells in the suprachiasmatic nucleus. Eur J Neurosci 12:571–576
Shibata S, Oomura Y, Kita H, Hattori K (1982) Circadian rhythmic changes of neuronal activity in the suprachiasmatic nucleus of the rat hypothalamic slice. Brain Res 247(1):154–158
Jiang ZG, Yang Y, Liu ZP, Allen CN (1997) Membrane properties and synaptic inputs of suprachiasmatic nucleus neurons in rat brain slices. J Physiol 499(Pt 1):141–159
Meijer JH, Schaap J, Watanabe K, Albus H (1997) Multiunit activity recordings in the suprachiasmatic nuclei: in vivo versus in vitro models. Brain Res 753(2):322–327
Schaap J, Bos NPA, deJeu MTG, Geurtsen AMS, Meijer JH, Pennartz CMA (1999) Neurons of the rat suprachiasmatic nucleus show a circadian rhythm in membrane properties that is lost during prolonged whole-cell recording. Brain Res 815(1):154–166
Jin H, Ham SA, Kim MY, Woo IS, Kang ES, Hwang JS, Lee KW, Kim HJ, Roh GS, Lim DS, Kang D, Seo HG (2012) Activation of peroxisome proliferator-activated receptor-delta attenuates glutamate-induced neurotoxicity in HT22 mouse hippocampal cells. J Neurosci Res 90(8):1646–1653
Nelson DE, Takahashi JS (1991) Sensitivity and integration in a visual pathway for circadian entrainment in the Hamster (Mesocricetus-Auratus). J Physiol 439:115–145
Muscat L, Morin LP (2005) Binocular contributions to the responsiveness and integrative capacity of the circadian rhythm system to light. J Biol Rhythms 20(6):513–525
Kallingal GJ, Mintz EM (2006) Glutamatergic activity modulates the phase-shifting effects of gastrin-releasing peptide and light. Eur J Neurosci 24(10):2853–2858
Glinghammar B, Skogsberg J, Hamsten A, Ehrenborg E (2003) PPARdelta activation induces COX-2 gene expression and cell proliferation in human hepatocellular carcinoma cells. Biochem Biophys Res Commun 308(2):361–368
Xu L, Han C, Wu T (2006) A novel positive feedback loop between peroxisome proliferator-activated receptor-delta and prostaglandin E2 signaling pathways for human cholangiocarcinoma cell growth. J Biol Chem 281(45):33982–33996
Yang H, Chen C (2008) Cyclooxygenase-2 in synaptic signaling. Curr Pharm Des 14(14):1443–1451
Chen C, Bazan NG (2005) Lipid signaling: sleep, synaptic plasticity, and neuroprotection. Prostaglandins Other Lipid Mediat 77(1–4):65–76
Sang N, Zhang J, Marcheselli V, Bazan NG, Chen C (2005) Postsynaptically synthesized prostaglandin E2 (PGE2) modulates hippocampal synaptic transmission via a presynaptic PGE2 EP2 receptor. J Neurosci 25(43):9858–9870
Bazan NG (2003) Synaptic lipid signaling: significance of polyunsaturated fatty acids and platelet-activating factor. J Lipid Res 44(12):2221–2233
Sanzgiri RP, Araque A, Haydon PG (1999) Prostaglandin E2 stimulates glutamate receptor-dependent astrocyte neuromodulation in cultured hippocampal cells. J Neurobiol 41(2):221–229
Bezzi P, Carmignoto G, Pasti L, Vesce S, Rossi D, Rizzini BL, Pozzan T, Volterra A (1998) Prostaglandins stimulate calcium-dependent glutamate release in astrocytes. Nature 391(6664):281–285
Dave KA, Platel J-C, Huang F, Tian D, Stamboulian-Platel S, Bordey A (2011) Prostaglandin E2 induces glutamate release from subventricular zone astrocytes. Neuron Glia Biol 7:1–7
Wagner K-D, Wagner N (2010) Peroxisome proliferator-activated receptor beta/delta (PPARbeta/delta) acts as regulator of metabolism linked to multiple cellular functions. Pharmacol Ther 125(3):423–435
Acknowledgments
The authors would like to thank Paul Pévet for helpful comments on the manuscript, Claire Maudet for animal care, Stéphanie Dumont for preparing drug solutions, Gaëlle Champeil-Potokar for confocal images. This work was supported by the Institut National de la Recherche Agronomique (INRA), Centre National de la Recherche Scientifique (CNRS) and University of Strasbourg. All authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Challet, E., Denis, I., Rochet, V. et al. The role of PPARβ/δ in the regulation of glutamatergic signaling in the hamster suprachiasmatic nucleus. Cell. Mol. Life Sci. 70, 2003–2014 (2013). https://doi.org/10.1007/s00018-012-1241-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-012-1241-9