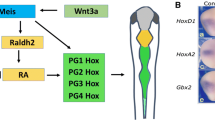
Abstract
The midbrain-hindbrain boundary (MHB) is a highly conserved vertebrate signalling centre, acting to pattern and establish neural identities within the brain. While the core signalling pathways regulating MHB formation have been well defined, novel genetic and mechanistic processes that interact with these core components are being uncovered, helping to further elucidate the complicated networks governing MHB specification, patterning and shaping. Although formation of the MHB organiser is traditionally thought of as comprising three stages, namely positioning, induction and maintenance, we propose that a fourth stage, morphogenesis, should be considered as an additional stage in MHB formation. This review will examine evidence for novel factors regulating the first three stages of MHB development and will explore the evidence for regulation of MHB morphogenesis by non-classical MHB-patterning genes.




Similar content being viewed by others
References
Hirth F (2010) On the origin and evolution of the tripartite brain. Brain Behav Evol 76(1):3–10
Lowery LA, Sive H (2009) Totally tubular: the mystery behind function and origin of the brain ventricular system. BioEssays: news and reviews in molecular, cellular and developmental biology 31(4):446–458
Echelard Y et al (1993) Sonic hedgehog, a member of a family of putative signaling molecules, is implicated in the regulation of CNS polarity. Cell 75(7):1417–1430
Lee KJ, Jessell TM (1999) The specification of dorsal cell fates in the vertebrate central nervous system. Ann Rev Neurosci 22:261–294
Marin F, Puelles L (1994) Patterning of the embryonic avian midbrain after experimental inversions: a polarizing activity from the isthmus. Dev Biol 163(1):19–37
Martinez S et al (1995) Induction of ectopic engrailed expression and fate change in avian rhombomeres: intersegmental boundaries as barriers. Mech Dev 51(2–3):289–303
Martinez S, Wassef M, Alvarado-Mallart RM (1991) Induction of a mesencephalic phenotype in the 2-day-old chick prosencephalon is preceded by the early expression of the homeobox gene en. Neuron 6(6):971–981
Alvarado-Mallart RM (2005) The chick/quail transplantation model: discovery of the isthmic organizer center, brain research. Brain Res Rev 49(2):109–113
Rhinn M, Brand M (2001) The midbrain–hindbrain boundary organizer. Curr Opinion Neurobiol 11(1):34–42
Joyner AL, Liu A, Millet S (2000) Otx2, Gbx2 and Fgf8 interact to position and maintain a mid-hindbrain organizer. Curr Opin Cell Biol 12(6):736–741
Broccoli V, Boncinelli E, Wurst W (1999) The caudal limit of Otx2 expression positions the isthmic organizer. Nature 401(6749):164–168
Simeone A (2000) Positioning the isthmic organizer where Otx2 and Gbx2meet. Trends Genetics : TIG 16(6):237–240
Brand M et al (1996) Mutations in zebrafish genes affecting the formation of the boundary between midbrain and hindbrain. Development 123:179–190
Picker A et al (2002) A novel positive transcriptional feedback loop in midbrain–hindbrain boundary development is revealed through analysis of the zebrafish pax2.1 promoter in transgenic lines. Development 129(13):3227–3239
Borello U, Pierani A (2010) Patterning the cerebral cortex: traveling with morphogens. Curr Opin Genet Dev 20(4):408–415
Wurst W, Bally-Cuif L (2001) Neural plate patterning: upstream and downstream of the isthmic organizer. Nat Rev Neurosci 2(2):99–108
Hidalgo-Sanchez M et al (2005) Specification of the meso-isthmo-cerebellar region: the Otx2/Gbx2 boundary. Brain Res Brain Res Rev 49(2):134–149
Barkovich AJ, Millen KJ, Dobyns WB (2009) A developmental and genetic classification for midbrain–hindbrain malformations. Brain J Neurol 132(Pt 12):3199–3230
Wallingford JB (2006) Planar cell polarity, ciliogenesis and neural tube defects. Hum Mol Genet 15(Spec No 2):R227–R234
Rhinn M et al (2005) Positioning of the midbrain–hindbrain boundary organizer through global posteriorization of the neuroectoderm mediated by Wnt8 signaling. Development 132(6):1261–1272
Klingensmith J et al (1999) Neural induction and patterning in the mouse in the absence of the node and its derivatives. Dev Biol 216(2):535–549
Liu A, Joyner AL (2001) EN and GBX2 play essential roles downstream of FGF8 in patterning the mouse mid/hindbrain region. Development 128(2):181–191
Sato T, Joyner AL (2009) The duration of Fgf8 isthmic organizer expression is key to patterning different tectal-isthmo-cerebellum structures. Development 136(21):3617–3626
Li JY, Joyner AL (2001) Otx2 and Gbx2 are required for refinement and not induction of mid-hindbrain gene expression. Development 128(24):4979–4991
Agoston Z, Schulte D (2009) Meis2 competes with the Groucho co-repressor Tle4 for binding to Otx2 and specifies tectal fate without induction of a secondary midbrain–hindbrain boundary organizer. Development 136(19):3311–3322
Jungbluth S et al (2001) Cell mixing between the embryonic midbrain and hindbrain. Curr Biol 11(3):204–207
Langenberg T, Brand M (2005) Lineage restriction maintains a stable organizer cell population at the zebrafish midbrain–hindbrain boundary. Development 132(14):3209–3216
Tallafuss A, Bally-Cuif L (2003) Tracing of her5 progeny in zebrafish transgenics reveals the dynamics of midbrain–hindbrain neurogenesis and maintenance. Development 130(18):4307–4323
Tossell K et al (2011) Lrrn1 is required for formation of the midbrain–hindbrain boundary and organiser through regulation of affinity differences between midbrain and hindbrain cells in chick. Dev Biol 352(2):341–352
Holland LZ, Short S (2008) Gene duplication, co-option and recruitment during the origin of the vertebrate brain from the invertebrate chordate brain. Brain Behav Evol 72(2):91–105
Glavic A, Gomez-Skarmeta JL, Mayor R (2002) The homeoprotein Xiro1 is required for midbrain–hindbrain boundary formation. Development 129(7):1609–1621
Crossley PH, Martinez S, Martin GR (1996) Midbrain development induced by FGF8 in the chick embryo. Nature 380(6569):66–68
Reifers F et al (1998) Fgf8 is mutated in zebrafish acerebellar (ace) mutants and is required for maintenance of midbrain–hindbrain boundary development and somitogenesis. Development 125(13):2381–2395
Leucht C et al (2008) MicroRNA-9 directs late organizer activity of the midbrain–hindbrain boundary. Nat Neurosci 11(6):641–648
Geling A et al (2003) bHLH transcription factor Her5 links patterning to regional inhibition of neurogenesis at the midbrain–hindbrain boundary. Development 130(8):1591–1604
Chi CL et al (2003) The isthmic organizer signal FGF8 is required for cell survival in the prospective midbrain and cerebellum. Development 130(12):2633–2644
Badde A, Schulte D (2008) A role for receptor protein tyrosine phosphatase lambda in midbrain development. J Neurosci Off J Soc Neurosci 28(24):6152–6164
Itoh M et al (2002) A role for iro1 and iro7 in the establishment of an anteroposterior compartment of the ectoderm adjacent to the midbrain–hindbrain boundary. Development 129(10):2317–2327
Tossell K et al (2011) Notch signalling stabilises boundary formation at the midbrain–hindbrain organiser. Development 138(17):3745–3757
Sugiyama S, Funahashi J, Nakamura H (2000) Antagonizing activity of chick Grg4 against tectum-organizing activity. Dev Biol 221(1):168–180
Shinga J et al (2001) Early patterning of the prospective midbrain–hindbrain boundary by the HES-related gene XHR1 in Xenopus embryos. Mech Dev 109(2):225–239
Canning CA et al (2007) Sustained interactive Wnt and FGF signaling is required to maintain isthmic identity. Dev Biol 305(1):276–286
Wittmann DM et al (2009) Spatial analysis of expression patterns predicts genetic interactions at the mid-hindbrain boundary. PLoS Comput Biol 5(11):e1000569
Dworkin S et al (2012) Midbrain–hindbrain boundary patterning and morphogenesis are regulated by diverse grainy head-like 2-dependent pathways. Development 139(3):525–536
Nakamura H, Sato T, Suzuki-Hirano A (2008) Isthmus organizer for mesencephalon and metencephalon. Dev Growth Differ 50(Suppl 1):S113–S118
Suzuki-Hirano A, Sato T, Nakamura H (2005) Regulation of isthmic Fgf8 signal by sprouty2. Development 132(2):257–265
Kim M, McGinnis W (2011) Phosphorylation of Grainy head by ERK is essential for wound-dependent regeneration but not for development of an epidermal barrier. Proc Nat Acad Sci USA 108(2):650–655
Buckles GR et al (2004) Combinatorial Wnt control of zebrafish midbrain–hindbrain boundary formation. Mech Dev 121(5):437–447
Adams KA et al (2000) The transcription factor Lmx1b maintains Wnt1 expression within the isthmic organizer. Development 127(9):1857–1867
O’Hara FP et al (2005) Zebrafish Lmx1b.1 and Lmx1b.2 are required for maintenance of the isthmic organizer. Development 132(14):3163–3173
Guo C et al (2007) Lmx1b is essential for Fgf8 and Wnt1 expression in the isthmic organizer during tectum and cerebellum development in mice. Development 134(2):317–325
Erickson T et al (2007) Pbx proteins cooperate with Engrailed to pattern the midbrain–hindbrain and diencephalic-mesencephalic boundaries. Developmental biology 301(2):504–517
Koike S et al (2011) Autotaxin is required for the cranial neural tube closure and establishment of the midbrain–hindbrain boundary during mouse development. Dev Dyn Off Publ Am Assoc Anat 240(2):413–421
Belting HG et al (2001) spiel ohne grenzen/pou2 is required during establishment of the zebrafish midbrain–hindbrain boundary organizer. Development 128(21):4165–4176
Bouchard M et al (2005) Identification of Pax2-regulated genes by expression profiling of the mid-hindbrain organizer region. Development 132(11):2633–2643
Xu J, Liu Z, Ornitz DM (2000) Temporal and spatial gradients of Fgf8 and Fgf17 regulate proliferation and differentiation of midline cerebellar structures. Development 127(9):1833–1843
Xu FX, Chye ML (1999) Expression of cysteine proteinase during developmental events associated with programmed cell death in brinjal. Plant J Cell Mol Biol 17(3):321–327
Panhuysen M et al (2004) Effects of Wnt1 signaling on proliferation in the developing mid-/hindbrain region. Mol Cell Neurosci 26(1):101–111
Sgado P et al (2006) Slow progressive degeneration of nigral dopaminergic neurons in postnatal Engrailed mutant mice. Proc Nat Acad Sci USA 103(41):15242–15247
Alavian KN et al (2009) Elevated P75NTR expression causes death of engrailed-deficient midbrain dopaminergic neurons by Erk1/2 suppression. Neural Dev 4:11
Radice GL et al (1997) Developmental defects in mouse embryos lacking N-cadherin. Dev Biol 181(1):64–78
Lele Z et al (2002) parachute/n-cadherin is required for morphogenesis and maintained integrity of the zebrafish neural tube. Development 129(14):3281–3294
Dworkin S et al (2007) CREB activity modulates neural cell proliferation, midbrain–hindbrain organization and patterning in zebrafish. Dev Biol 307(1):127–141
Giraldez AJ et al (2005) MicroRNAs regulate brain morphogenesis in zebrafish. Science 308(5723):833–838
Gutzman JH et al (2008) Formation of the zebrafish midbrain–hindbrain boundary constriction requires laminin-dependent basal constriction. Mech Dev 125(11–12):974–983
Lowery LA, Sive H (2005) Initial formation of zebrafish brain ventricles occurs independently of circulation and requires the nagie oko and snakehead/atp1a1a.1 gene products. Development 132(9):2057–2067
Pirone DM, Fukuhara S, Gutkind JS, Burbelo PD (2000) SPECs, small binding proteins for Cdc42. J Biol Chem 275(30):22650–22656
Ching KH, Kisailus AE, Burbelo PD (2005) The role of SPECs, small Cdc42-binding proteins, in F-actin accumulation at the immunological synapse. J Biol Chem 280(25):23660–23667
Ching KH, Kisailus AE, Burbelo PD (2007) Biochemical characterization of distinct regions of SPEC molecules and their role in phagocytosis. Exp Cell Res 313(1):10–21
Mullins MC et al (1994) Large-scale mutagenesis in the zebrafish: in search of genes controlling development in a vertebrate. Curr Biol 4(3):189–202
Kudoh T et al (2001) A gene expression screen in zebrafish embryogenesis. Genome Res 11(12):1979–1987
Thisse B, Thisse C (2004) Fast Release Clones: A High Throughput Expression Analysis. ZFIN Direct Data Submission (http://zfin.org)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dworkin, S., Jane, S.M. Novel mechanisms that pattern and shape the midbrain-hindbrain boundary. Cell. Mol. Life Sci. 70, 3365–3374 (2013). https://doi.org/10.1007/s00018-012-1240-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-012-1240-x