Abstract
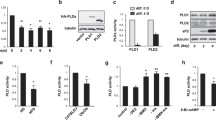
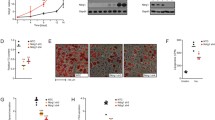
The role of protein kinase C (PKC) isoforms in the commitment of multipotent fibroblasts to the adipocyte lineage and in their terminal differentiation into mature adipocytes was investigated. Ectopic overexpression of PKC-ε, but not other PKC isoforms, committed multipotent NIH-3T3 cells to adipogenic differentiation in the presence of hormonal inducers. In committed 3T3-F442A preadipocytes, PKC-ε protein expression increased during the course of terminal differentiation and cell-permeable PKC-ε inhibitory peptides, which prevent interaction with RACK (receptor for activated C-kinase) proteins, severely inhibited differentiation. PKC-ε accumulated in the nuclei of 3T3-F442A cells shortly after induction of differentiation and exhibited a distinctive punctate speckling immunocytochemical staining pattern. The spatiotemporal aspects of PKC-ε localization and expression coincided with that of C/EBP-β, a transcription factor critically involved in promoting the early phase of adipogenesis. Collectively, these results demonstrate a role for PKC-ε in both adipogenic commitment and preadipocyte terminal differentiation.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Additional information
Received 28 November 2002; received after revision 25 February 2003; accepted 25 April 2003
Rights and permissions
About this article
Cite this article
Webb, P.R., Doyle, C. & Anderson, N.G. Protein kinase C-ε promotes adipogenic commitment and is essential for terminal differentiation of 3T3-F442A preadipocytes. CMLS, Cell. Mol. Life Sci. 60, 1504–1512 (2003). https://doi.org/10.1007/s00018-003-2337-z
Issue Date:
DOI: https://doi.org/10.1007/s00018-003-2337-z