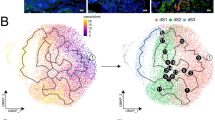
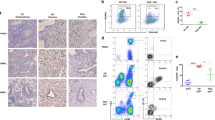
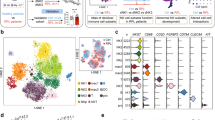
Abstract
Objective and design
To investigate the balancing mechanisms between decidualization-associated inflammation and pregnancy-related immunotolerance.
Material or subjects
Decidual samples from women with normal pregnancy (n = 58) or unexplained spontaneous miscarriage (n = 13), peripheral blood from normal pregnancy and endometria from non-pregnancy (n = 10) were collected. Primary endometrial stromal cells (ESCs), decidual stromal cells (DSCs), decidual immune cells (DICs) and peripheral blood mononuclear cells (PBMCs) were isolated.
Treatment
The plasmid carrying neuropilin-1 (NRP1) gene was transfected into ESC for overexpression. To induce decidualization in vitro, ESCs were treated with a combination of 10 nM estradiol, 100 nM progesterone and 0.5 mM cAMP. Anti-Sema3a and anti-NRP1 neutralizing antibodies were applied to block the ligand–receptor interactions.
Methods
RNA-seq analysis was performed to identify differentially expressed genes in DSCs and DICs, and NRP1 expression was verified by Western blotting and flow cytometry. The secretion of inflammatory mediators was measured using a multifactor cytometric bead array. The effects of Sema3a-NRP1 pathway on DICs were determined by flow cytometry. Statistical differences between groups were compared using the T test and one way or two-way ANOVA.
Results
Combined with five RNA-seq datasets, NRP1 was the only immune checkpoint changing oppositely between DSCs and DICs. The decreased expression of NRP1 in DSCs allowed intrinsic inflammatory responses required for decidualization, while its increased expression in DICs enhanced tolerant phenotypes beneficial to pregnancy maintenance. DSC-secreted Sema3a promoted immunosuppression in DICs via NRP1 binding. In women with miscarriage, NRP1 was abnormally elevated in DSCs but diminished in decidual macrophages and NK cells.
Conclusion
NRP1 is a multifunctional controller that balances the inflammatory states of DSCs and DICs in gravid uterus. Abnormal expression of NRP1 is implicated in miscarriage.








Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files. Further inquiries can be directed to the corresponding author on reasonable request.
References
Larsen EC, Christiansen OB, Kolte AM, Macklon N. New insights into mechanisms behind miscarriage. BMC Med. 2013;11:154. https://doi.org/10.1186/1741-7015-11-154.
Quenby S, Gallos ID, Dhillon-Smith RK, Podesek M, Stephenson MD, Fisher J, et al. Miscarriage matters: the epidemiological, physical, psychological, and economic costs of early pregnancy loss. Lancet (London, England). 2021;397(10285):1658–67. https://doi.org/10.1016/S0140-6736(21)00682-6.
Ander SE, Diamond MS, Coyne CB. Immune responses at the maternal-fetal interface. Sci Immunol. 2019. https://doi.org/10.1126/sciimmunol.aat6114.
Vento-Tormo R, Efremova M, Botting RA, Turco MY, Vento-Tormo M, Meyer KB, et al. Single-cell reconstruction of the early maternal-fetal interface in humans. Nature. 2018;563(7731):347–53. https://doi.org/10.1038/s41586-018-0698-6.
Ng S-W, Norwitz GA, Pavlicev M, Tilburgs T, Simón C, Norwitz ER. Endometrial decidualization: the primary driver of pregnancy health. Int J Mol Sci. 2020. https://doi.org/10.3390/ijms21114092.
Gellersen B, Brosens JJ. Cyclic decidualization of the human endometrium in reproductive health and failure. Endocr Rev. 2014;35(6):851–905. https://doi.org/10.1210/er.2014-1045.
Murakami K, Kuroda K, Brosens JJ. Perturbation of endometrial decidualization. In: Treatment strategy for unexplained infertility and recurrent miscarriage. Singapore: Springer; 2018. p. 105–14.
Zhang T, Zhu W, Zhao Y, Cheung WC, Liu Y, Chen X, et al. Early transient suppression of immune checkpoint proteins T-cell immunoglobulin mucin-3 and programmed cell death-1 in peripheral blood lymphocytes after blastocyst transfer is associated with successful implantation. Fertil Steril. 2020;114(2):426–35. https://doi.org/10.1016/j.fertnstert.2019.12.022.
Quenby S. Implantation involves an initial pro-inflammatory response. Fertil Steril. 2020;114(2):288–9. https://doi.org/10.1016/j.fertnstert.2020.05.039.
Erlebacher A. Immunology of the maternal-fetal interface. Annu Rev Immunol. 2013;31(1):387–411. https://doi.org/10.1146/annurev-immunol-032712-100003.
Nadeau-Vallée M, Obari D, Palacios J, Brien M-È, Duval C, Chemtob S, et al. Sterile inflammation and pregnancy complications: a review. Reproduction (Cambridge, England). 2016;152(6):R277–92.
Zhao S-J, Muyayalo KP, Luo J, Huang D, Mor G, Liao A-H. Next generation of immune checkpoint molecules in maternal-fetal immunity. Immunol Rev. 2022;308(1):40–54. https://doi.org/10.1111/imr.13073.
Wang S, Zhu X, Xu Y, Zhang D, Li Y, Tao Y, et al. Programmed cell death-1 (PD-1) and T-cell immunoglobulin mucin-3 (Tim-3) regulate CD4+ T cells to induce Type 2 helper T cell (Th2) bias at the maternal-fetal interface. Human Reprod (Oxford, England). 2016;31(4):700–11. https://doi.org/10.1093/humrep/dew019.
Li Y, Zhang J, Zhang D, Hong X, Tao Y, Wang S, et al. Tim-3 signaling in peripheral NK cells promotes maternal-fetal immune tolerance and alleviates pregnancy loss. Sci Signal. 2017. https://doi.org/10.1126/scisignal.aah4323.
Wang S, Sun F, Li M, Qian J, Chen C, Wang M, et al. The appropriate frequency and function of decidual Tim-3CTLA-4CD8 T cells are important in maintaining normal pregnancy. Cell Death Dis. 2019;10(6):407. https://doi.org/10.1038/s41419-019-1642-x.
Li M, Sun F, Qian J, Chen L, Li D, Wang S, et al. Tim-3/CTLA-4 pathways regulate decidual immune cells-extravillous trophoblasts interaction by IL-4 and IL-10. FASEB J. 2021;35(8): e21754. https://doi.org/10.1096/fj.202100142R.
He X, Xu C. Immune checkpoint signaling and cancer immunotherapy. Cell Res. 2020;30(8):660–9. https://doi.org/10.1038/s41422-020-0343-4.
Mohamed Khosroshahi L, Parhizkar F, Kachalaki S, Aghebati-Maleki A, Aghebati-Maleki L. Immune checkpoints and reproductive immunology: pioneers in the future therapy of infertility related Disorders? Int Immunopharmacol. 2021;99:1079. https://doi.org/10.1016/j.intimp.2021.107935.
Thompson KE, Danberry TL, Bunch RT, Graziano MJ, McNerney ME. Allogeneic murine pregnancy models for assessing the developmental effects of immune-stimulating antibodies: challenges in reproducibility. Birth Defects Res. 2019;111(16):1178–91. https://doi.org/10.1002/bdr2.1542.
Wang S, Chen C, Li M, Qian J, Sun F, Li Y, et al. Blockade of CTLA-4 and Tim-3 pathways induces fetal loss with altered cytokine profiles by decidual CD4T cells. Cell Death Dis. 2019;10(1):15. https://doi.org/10.1038/s41419-018-1251-0.
Wang S, Cao C, Piao H, Li Y, Tao Y, Zhang X, et al. Tim-3 protects decidual stromal cells from toll-like receptor-mediated apoptosis and inflammatory reactions and promotes Th2 bias at the maternal-fetal interface. Sci Rep. 2015;5:9013. https://doi.org/10.1038/srep09013.
Fu W, Cai R, Ma Z, Li T, Lei C, Zhao J, et al. TIGIT-Fc as a potential therapeutic agent for fetomaternal tolerance. Front Immunol. 2021;12:649135. https://doi.org/10.3389/fimmu.2021.649135.
Olivares EG, Montes MJ, Oliver C, Galindo JA, Ruiz C. Cultured human decidual stromal cells express B7–1 (CD80) and B7–2 (CD86) and stimulate allogeneic T cells. Biol Reprod. 1997;57(3):609–15.
Nagamatsu T, Schust DJ, Sugimoto J, Barrier BF. Human decidual stromal cells suppress cytokine secretion by allogenic CD4+ T cells via PD-1 ligand interactions. Hum Reprod (Oxford, England). 2009;24(12):3160–71. https://doi.org/10.1093/humrep/dep308.
Tao Y, Li Y-H, Zhang D, Xu L, Chen J-J, Sang Y-F, et al. Decidual CXCR4 CD56 NK cells as a novel NK subset in maternal-foetal immune tolerance to alleviate early pregnancy failure. Clin Transl Med. 2021;11(10): e540. https://doi.org/10.1002/ctm2.540.
Lv S, Liu M, Xu L, Zhang C. Downregulation of decidual SKP2 is associated with human recurrent miscarriage. Reprod Biol Endocrinol. 2021;19(1):88. https://doi.org/10.1186/s12958-021-00775-4.
Xu L, Li YH, Zhao WJ, Sang YF, Chen JJ, Li DJ, et al. RhoB promotes endometrial stromal cells decidualization via semaphorin3A/plexinA4 signaling in early pregnancy. Endocrinology. 2022. https://doi.org/10.1210/endocr/bqac134.
Roy S, Bag AK, Singh RK, Talmadge JE, Batra SK, Datta K. Multifaceted role of neuropilins in the immune system: potential targets for immunotherapy. Front Immunol. 2017;8:1228. https://doi.org/10.3389/fimmu.2017.01228.
Wang SC, Li YH, Piao HL, Hong XW, Zhang D, Xu YY, et al. PD-1 and Tim-3 pathways are associated with regulatory CD8+ T-cell function in decidua and maintenance of normal pregnancy. Cell Death Dis. 2015;6: e1738. https://doi.org/10.1038/cddis.2015.112.
Wherry EJ, Kurachi M. Molecular and cellular insights into T cell exhaustion. Nat Rev Immunol. 2015;15(8):486–99. https://doi.org/10.1038/nri3862.
Nancy P, Tagliani E, Tay C-S, Asp P, Levy DE, Erlebacher A. Chemokine gene silencing in decidual stromal cells limits T cell access to the maternal-fetal interface. Science (New York, NY). 2012;336(6086):1317–21. https://doi.org/10.1126/science.1220030.
Du L, Deng W, Zeng S, Xu P, Huang L, Liang Y, et al. Single-cell transcriptome analysis reveals defective decidua stromal niche attributes to recurrent spontaneous abortion. Cell Prolif. 2021. https://doi.org/10.1111/cpr.13125.
Zhang X, Wei H. Role of decidual natural killer cells in human pregnancy and related pregnancy complications. Front Immunol. 2021;12:728291. https://doi.org/10.3389/fimmu.2021.728291.
Piccinni M-P, Raghupathy R, Saito S, Szekeres-Bartho J. Cytokines, hormones and cellular regulatory mechanisms favoring successful reproduction. Front Immunol. 2021;12:717808. https://doi.org/10.3389/fimmu.2021.717808.
Wang W, Sung N, Gilman-Sachs A, Kwak-Kim J. T Helper (Th) cell profiles in pregnancy and recurrent pregnancy losses: Th1/Th2/Th9/Th17/Th22/Tfh cells. Front Immunol. 2020;11:2025. https://doi.org/10.3389/fimmu.2020.02025.
Delgoffe GM, Woo S-R, Turnis ME, Gravano DM, Guy C, Overacre AE, et al. Stability and function of regulatory T cells is maintained by a neuropilin-1-semaphorin-4a axis. Nature. 2013;501(7466):252–6. https://doi.org/10.1038/nature12428.
Mekinian A, Cohen J, Alijotas-Reig J, Carbillon L, Nicaise-Roland P, Kayem G, et al. Unexplained recurrent miscarriage and recurrent implantation failure: is there a place for immunomodulation? Am J Reprod Immunol (New York, NY : 1989). 2016. https://doi.org/10.1111/aji.12493.
Chavan AR, Griffith OW, Wagner GP. The inflammation paradox in the evolution of mammalian pregnancy: turning a foe into a friend. Curr Opin Genet Dev. 2017;47:24–32. https://doi.org/10.1016/j.gde.2017.08.004.
Kitsukawa T, Shimono A, Kawakami A, Kondoh H, Fujisawa H. Overexpression of a membrane protein, neuropilin, in chimeric mice causes anomalies in the cardiovascular system, nervous system and limbs. Development. 1995;121(12):4309–18.
Kawasaki T, Kitsukawa T, Bekku Y, Matsuda Y, Sanbo M, Yagi T, et al. A requirement for neuropilin-1 in embryonic vessel formation. Development. 1999;126(21):4895–902.
Yuan L, Moyon D, Pardanaud L, Bréant C, Karkkainen MJ, Alitalo K, et al. Abnormal lymphatic vessel development in neuropilin 2 mutant mice. Development. 2002;129(20):4797–806.
Huang Z, Huang S, Song T, Yin Y, Tan C. Placental angiogenesis in mammals: a review of the regulatory effects of signaling pathways and functional nutrients. Adv Nutr. 2021;12(6):2415–34. https://doi.org/10.1093/advances/nmab070.
Arad A, Nammouz S, Nov Y, Ohel G, Bejar J, Vadasz Z. The expression of neuropilin-1 in human placentas from normal and preeclamptic pregnancies. Int J Gynecol Pathol. 2017;36(1):42–9. https://doi.org/10.1097/PGP.0000000000000283.
Baston-Buest DM, Porn AC, Schanz A, Kruessel J-S, Janni W, Hess AP. Expression of the vascular endothelial growth factor receptor neuropilin-1 at the human embryo-maternal interface. Eur J Obstet Gynecol Reprod Biol. 2011;154(2):151–6. https://doi.org/10.1016/j.ejogrb.2010.10.018.
Maulik D, De A, Ragolia L, Evans J, Grigoryev D, Lankachandra K, et al. Down-regulation of placental neuropilin-1 in fetal growth restriction. Am J Obstet Gynecol. 2016;214(2):2791–9. https://doi.org/10.1016/j.ajog.2015.09.068.
Yang X, Chen D, He B, Cheng W. NRP1 and MMP9 are dual targets of RNA-binding protein QKI5 to alter VEGF-R/ NRP1 signalling in trophoblasts in preeclampsia. J Cell Mol Med. 2021;25(12):5655–70. https://doi.org/10.1111/jcmm.16580.
Xu X, Yang XY, He BW, Yang WJ, Cheng WW. Placental NRP1 and VEGF expression in pre-eclamptic women and in a homocysteine-treated mouse model of pre-eclampsia. Eur J Obstet Gynecol Reprod Biol. 2016;196:69–75. https://doi.org/10.1016/j.ejogrb.2015.11.017.
Zhou Y, Gormley MJ, Hunkapiller NM, Kapidzic M, Stolyarov Y, Feng V, et al. Reversal of gene dysregulation in cultured cytotrophoblasts reveals possible causes of preeclampsia. J Clin Investig. 2013;123(7):2862–72. https://doi.org/10.1172/JCI66966.
Chuckran CA, Liu C, Bruno TC, Workman CJ, Vignali DA. Neuropilin-1: a checkpoint target with unique implications for cancer immunology and immunotherapy. J Immunother Cancer. 2020. https://doi.org/10.1136/jitc-2020-000967.
Weiss JM, Bilate AM, Gobert M, Ding Y, de Curotto Lafaille MA, Parkhurst CN, et al. Neuropilin 1 is expressed on thymus-derived natural regulatory T cells, but not mucosa-generated induced Foxp3+ T reg cells. J Exp Med. 2012;209(10):1723–42.
Jackson SR, Berrien-Elliott M, Yuan J, Hsueh EC, Teague RM. Neuropilin-1 expression is induced on tolerant self-reactive CD8+ T cells but is dispensable for the tolerant phenotype. PLoS ONE. 2014;9(10): e110707. https://doi.org/10.1371/journal.pone.0110707.
Sarris M, Andersen KG, Randow F, Mayr L, Betz AG. Neuropilin-1 expression on regulatory T cells enhances their interactions with dendritic cells during antigen recognition. Immunity. 2008;28(3):402–13. https://doi.org/10.1016/j.immuni.2008.01.012.
Miyauchi JT, Caponegro MD, Chen D, Choi MK, Li M, Tsirka SE. Deletion of neuropilin 1 from microglia or bone marrow-derived macrophages slows glioma progression. Can Res. 2018;78(3):685–94. https://doi.org/10.1158/0008-5472.CAN-17-1435.
Wallerius M, Wallmann T, Bartish M, Östling J, Mezheyeuski A, Tobin NP, et al. Guidance molecule SEMA3A restricts tumor growth by differentially regulating the proliferation of tumor-associated macrophages. Can Res. 2016;76(11):3166–78. https://doi.org/10.1158/0008-5472.CAN-15-2596.
Grasso E, Gori S, Soczewski E, Fernández L, Gallino L, Vota D, et al. Impact of the reticular stress and unfolded protein response on the inflammatory response in endometrial stromal cells. Sci Rep. 2018;8(1):12274. https://doi.org/10.1038/s41598-018-29779-8.
Mor G, Cardenas I, Abrahams V, Guller S. Inflammation and pregnancy: the role of the immune system at the implantation site. Ann N Y Acad Sci. 2011;1221:80–7. https://doi.org/10.1111/j.1749-6632.2010.05938.x.
Plaks V, Birnberg T, Berkutzki T, Sela S, BenYashar A, Kalchenko V, et al. Uterine DCs are crucial for decidua formation during embryo implantation in mice. J Clin Investig. 2008;118(12):3954–65. https://doi.org/10.1172/JCI36682.
Barash A, Dekel N, Fieldust S, Segal I, Schechtman E, Granot I. Local injury to the endometrium doubles the incidence of successful pregnancies in patients undergoing in vitro fertilization. Fertil Steril. 2003;79(6):1317–22.
Zhu Y-Y, Wu Y, Chen S-T, Kang J-W, Pan J-M, Liu X-Z, et al. Synthesized monosodium urate crystal enhances endometrium decidualization via sterile inflammation during pregnancy. Front Cell Dev Biol. 2021;9:702590. https://doi.org/10.3389/fcell.2021.702590.
Salker MS, Nautiyal J, Steel JH, Webster Z, Sućurović S, Nicou M, et al. Disordered IL-33/ST2 activation in decidualizing stromal cells prolongs uterine receptivity in women with recurrent pregnancy loss. PLoS ONE. 2012;7(12): e52252. https://doi.org/10.1371/journal.pone.0052252.
Gu X-W, Chen Z-C, Yang Z-S, Yang Y, Yan Y-P, Liu Y-F, et al. Blastocyst-induced ATP release from luminal epithelial cells initiates decidualization through the P2Y2 receptor in mice. Sci Signal. 2020. https://doi.org/10.1126/scisignal.aba3396.
Chavan AR, Griffith OW, Stadtmauer DJ, Maziarz J, Pavlicev M, Fishman R, et al. Evolution of embryo implantation was enabled by the origin of decidual stromal cells in eutherian mammals. Mol Biol Evol. 2021;38(3):1060–74. https://doi.org/10.1093/molbev/msaa274.
Acknowledgements
We thank all the patients who participated in our study. We are also grateful to all the members in our laboratory for their help.
Funding
This work was supported by the grants from the National Natural Science Foundation of China (82271713, 31970859, 81630036, 91542116, 31570920, 32070915, 31900663), National Science and Technology Program during the 13th Five-year Plan Period (2021YFE0206500), International cooperation project between Macao and Shanghai Municipal Commission of science and technology (20410760300), Strategic Collaborative Research Program of the Ferring Institute of Reproductive Medicine (FIRMA200504).
Author information
Authors and Affiliations
Contributions
JC and YL collected human samples, performed experiments, analyzed the data and wrote the manuscript. LX and YS generated figures, searched for literature and revised the manuscript. DL conceived the whole research and monitored the execution of this study strictly. MD designed the experiments, supplemented key materials, conducted data interpretation and revised the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
This study was approved by the Research Ethics Committee of the Obstetrics and Gynecology Hospital, Fudan university. Written informed consents were obtained from all subjects for the collection and study of their tissue or blood samples.
Additional information
Responsible Editor: John Di Battista.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, J., Li, Y., Xu, L. et al. Paradoxical expression of NRP1 in decidual stromal and immune cells reveals a novel inflammation balancing mechanism during early pregnancy. Inflamm. Res. 72, 1341–1357 (2023). https://doi.org/10.1007/s00011-023-01734-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-023-01734-y