Abstract
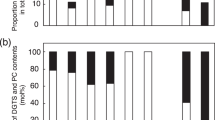
A mutant of the red microalga Porphyridium cruentum was selected on the basis of impaired growth at suboptimal temperatures (15 vs. 25°C). Fatty acid and lipid analyses revealed diminished proportions of eicosapentaenoic acid (from 41 to 30%) and of the eukaryotic molecular species (from 38 to 28% of monogalactosyldiacylglycerol (MGDG) and elevated proportion (10 vs. 2%) of triacylglycerols (TAG) in the mutant, as compared with the wild type. Pulse labeling of the wild type cells with radioactive fatty acid precursors indicated an initial incorporation of the fatty acids into phosphatidylcholine (PC) and TAG. Following the pulse, the label of PC and TAG decreased with time (from 25 to 5% of the total dpm in TAG) while that of chloroplastic polar lipids, mainly MGDG, continued to increase. In the mutant, however, the labeling of TAG after the pulse was higher (30% of the total dpm) than that of the wild type and decreased only slightly to 20%. This may indicate that in P. cruentum, TAG can contribute to the biosynthesis of eukaryotic species of MGDG.
Similar content being viewed by others
Abbreviations
- AA:
-
arachidonic acid
- C n :
-
fatty acid, fatty acid with n carbon atoms
- DAG:
-
diacylglycerol
- DAGAT:
-
diacylglycerol acyltransferase
- DGDG:
-
digalactosyldiacylglycerol
- EPA:
-
elcosapentaenoic acid (20∶5n−3)
- MGDG:
-
monogalactosyldiacylglycerol
- PC:
-
phosphatidylcholine
- PUFA:
-
polyunsaturated fatty acid
- TAG:
-
triacylglycerol
- WT:
-
wild type
- X:Y:
-
a fatty acyl group containing X carbon atoms and Y double bonds (cis). Pairs of numbers representing the fatty acids, when separated by a slash, designate the components in the sn-1 and sn-2 positions, respectively, of the molecular species
References
Browse, J., and Somerville, C.R. (1991) Glycerolipid Synthesis: Biochemistry and Regulation, Annu. Rev. Plant Mol. Biol. 42, 467–506.
Norman, H.A., Smith, L.A., Lynch, D.V., and Thompson, G.A. (1985) Effects of Low-Temperature Stress on the Metabolism of Phosphatidylglycerol Molecular Species in Dunaliella salina, Arch. Biochem. Biophys. 24, 157–167.
Arao, T., Sakaki, T., and Yamada, M. (1994) Biosynthesis of Polyunsaturated Lipids in the Diatom, Phaeodactylum tricornutum, Phytochemistry 36, 629–635.
Arao, T., and Yamada, M. (1994) Biosynthesis of Polyunsaturated Fatty Acids in the Marine Diatom, Phaeodactylum tricornutum, Phytochemistry 35, 1177–1181.
Cohen, Z., Norman, H.A., and Heimer, Y.M. (1995) Microalgae as a Source of Omega-3 Fatty Acids, in Plants in Human Nutrition, World Review of Nutrition and Dietetics (Simopoulos A.P., ed.) Vol. 77, pp. 1–31, Karger, Basel.
Simopoulos, A.P. (1991) ω-3 Fatty Acids in Health and Disease and in Growth and Development, Am. J. Clin. Nutr. 54, 438–463.
Shiran, D., Khozin, I., Heimer, Y.M., and Cohen, Z. (1996) Elucidation of the Biosynthesis of EPA in Porphyridium cruentum. I: The Use of Externally Supplied Fatty Acids, Lipids 31, 1277–1282.
Khozin, I., Adlerstein, D., Bigogno, C., Heimer, Y.M., and Cohen, Z. (1997) Elucidation of the Biosynthesis of EPA in the Microalga Porphyridium cruentum II: Radiolabeling Studies, Plant Physiol. 114, 223–230.
Schneider, J.C., Livne, A., Sukenik, A., and Roessler, P.G. (1995) A Mutant of Nannochloropsis Deficient in Eicosapentaenoic Acid Production, Phytochemistry 40, 807–814.
Wada, H., Gombos, Z., Sakamoto, T., and Murata, N. (1992) Genetic Manipulation of the Extent of Desaturation of Fatty Acids in Membrane Lipids in the Cyanobacterium Synechocystis PCC6803, Plant Cell Physiol. 33, 535–540.
Cohen, Z., Vonshak, A., and Richmond, A. (1988) Effect of Environmental Conditions of Fatty Acid Composition of the Red Alga Porphyridium cruentum: Correlation to Growth Rate, J. Phycol. 24, 328–332.
Wada, H., and Murata, N. (1989) Synechocystic PCC 6803, Mutants Defective in Desaturation of Fatty Acids, Plant Cell Physiol. 30, 971–978.
Jones, R.E., Speer, L., and Kury, W. (1983) Studies on the Growth of the Red Alga Porphyridium cruentum, Physiol. Plant. 16, 636–643.
Bligh, E.G., and Dyer, W.J. (1959) A Rapid Method for Total Lipid Extraction and Purification, Can. J. Biochem. 37, 911–917.
Lynch, D.V., Gundersen, R.E., and Thompson, G.A. (1983) Separation of Galactolipid Molecular Species by High Performance Liquid Chromatography, Plant Physiol. 72, 903–905.
Cohen, Z. (1990) The Production Potential of Eicosapentaenoic Acid and Arachidonic Acid of the Red Alga Porphyridium cruentum, J. Am. Oil Chem. Soc. 67, 916–920.
Adlerstein, D., Khozin, I., Bigogno, C., and Cohen, Z. (1997) The Effect of Growth Temperature and Culture Density on the Molecular Species Composition of the Galactolipids in the Red Microalga Porphyridium cruentum (Rhodophyta), J. Phycol. 33, 975–979.
Miquel, M., James, D., Dooner, J., and Browse, J. (1993) Arabidopsis Requires Polyunsaturated Lipids for Low-temperature Survival, Proc. Natl. Acad. Sci. U.S.A. 90, 6208–6212.
Wada, H., Gombos, Z., and Murata, N. (1990) Enhancement of Chilling Tolerance of a Cyanobacterium by Genetic Manipulation of Fatty Acid Desaturation, Nature 347, 200–203.
Garces, R., Sarmiento, C., and Mancha, M. (1994) Oleate from Triacylglycerols Is Desaturated in Cold-induced Developing Sunflower (Helianthus annuus L.) Seeds, Planta 193, 473–477.
Stobart, K., Mancha, M., Lenman, M., Dahlqvist, A., and Stymne, S. (1997) Triacylglycerols are Synthesized and Utilized by Transacylation Reactions in Microsomal Preparations of Developing Safflower (Carthamus tinctorius L.) Seeds, Planta 203, 58–66.
Henderson, R.J., Hodgson, P., and Harwood, J.L. (1990) Differential Effects of the Substituted Pyridazinone Herbicide Sandoz 9785 on Lipid Composition and Biosynthesis in Photosynthetic and Non-photosynthetic Marine Microalgae. II. Fatty Acid Composition, J. Exp. Bot. 41, 729–736.
Sukenik, A., and Carmeli, Y. (1990) Lipid Synthesis and Fatty Acid Composition in Nannochloropsis sp. (Eustimatophyceae) Grown in a Light-Dark Cycle, J. Phyc. 26, 464–469.
Cohen, Z. (1994) Production Potential of Eicosapentaenoic Acid by Monodus subterraneus, J. Am. Oil Chem. Soc. 71, 941–945.
Yongmanitchai, W., and Ward, O.P. (1991) Growth of and Omega-3 Fatty Acid Production by Phaeodactylum tricornutum Under Different Culture Conditions, Appl. Environ. Microbiol. 57, 419–425.
Henderson, R.J., and McKinley, E.E. (1992) Radiolabeling Studies of Lipids in the Marine Cryptomonad Chroomonas salina in Relation to the Fatty Acid Desaturation, Plant Cell Physiol. 33, 395–406.
Makewicz, A., Gribi, C., and Eichenberger, W. (1997) Lipids of Ectocarpus fasciculatus (Phaeophyceae). Incorporation of [1-14C]Oleate and the Role of TAG and MGDG in Lipid Metabolism, Plant Cell Physiol. 38, 952–60.
Eichenberger, W., and Gribi, C. (1997) Lipids of Pavlova lutheri: Cellular Site and Metabolic Role of DGCC, Phytochemistry 45, 1561–1567.
Falk-Peterson, S., Sargent, J.R., Hegseth, E.N., Hop, H., and Oklodkov, Y.B. (1998) Lipid and Fatty Acid Composition in Ice Algae and Phytoplankton from the Marginal Ice Zone in the Barents Sea, Polar Biology 20, 41–47.
Pettitt, T.R., Jones, A.L., and harwood, J.L. (1989) Lipids of the Marine Red Algae, Chondrus crispus and Polysiphonia lanosa, Phytochemistry 28, 2053–2058.
Araki, S., Sakurai, T., Oohusa, T., Kayama, M., and Nizsizawa, K. (1990) Content of Arachidonic and Eicosapentaenoic Acids in Polar Lipid from Gracilaria (Gracilariales, Rhodophyta), Hydrobiologia 204/205, 513–519.
Browse, J., and Somerville, C.R. (1994) Glycerolipids, in Arabidopsis (Meyerowitz, E.M., and Sormerville, C.R., eds.) pp. 881–912, Cold Spring Harbor Laboratory Press, Plainview.
Klyachko-Gurvich, G., Tsoglin, L.N., Doucha, J., Kopetskii, J., Shebalina, B.I., and Semenenko, V.E. (1999) Desaturation of Fatty Acids as an Adaptive Response to Shifts in Light Intensity, Physiol. Plant. 107, 240–249.
Wanner, G., and Kost, H.P. (1984) “Membrane Storage” of the Red Alga Porphyridium cruentum During Nitrate and Sulfate Starvation, Z. Pflanzenphysiol. 113, 251–262.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Khozin-Goldberg, I., Yu, H.Z., Adlerstein, D. et al. Triacylglycerols of the red microalga Porphyridium cruentum can contribute to the biosynthesis of eukaryotic galactolipids. Lipids 35, 881–889 (2000). https://doi.org/10.1007/S11745-000-0597-8
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/S11745-000-0597-8