Abstract:
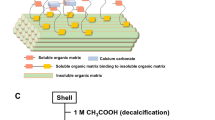
Calcified shell layer is composed of two polymorphs of CaCO3, aragonite or calcite, and an organic matrix. The organic matrix consists of EDTA-soluble and insoluble fractions. These fractions are thought to regulate the formation of the elaborate shell structure. After decalcification of powdered pearl with 0.3 M EDTA, an EDTA-insoluble fraction was extracted with 0.3 M EDTA/8 M urea. This extraction step enabled us to purify a new class of EDTA-insoluble protein, Pearlin, almost homogeneously. Pearlin has a molecular weight of about 15 kDa and contains a sulfated mucopolysaccharide. We cloned the complementary DNA coding for Pearlin and deduced its complete amino acid sequence. Sequence analysis reveals that Pearlin is composed of 129 amino acids with a high proportion of Gly (10.8%), Tyr (10.0%), Cys (8.5%), Asn (7.7%), Asp (7.7%), and Arg (7.7%). Northern blot analysis showed that Pearlin messenger RNA was expressed specifically in mantle epithelium. From the sequencing data, Pearlin was shown to be quite different from the fibrous protein rich in Ala and Gly. The function of this protein in biomineralization is discussed.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received May 5, 1999; accepted February 11, 2000.
Rights and permissions
About this article
Cite this article
Miyashita, T., Takagi, R., Okushima, M. et al. Complementary DNA Cloning and Characterization of Pearlin, a New Class of Matrix Protein in the Nacreous Layer of Oyster Pearls. Mar. Biotechnol. 2, 409–418 (2000). https://doi.org/10.1007/PL00021687
Issue Date:
DOI: https://doi.org/10.1007/PL00021687