Abstract
Background. This phase II study was designed to determine the toxicity and efficacy of a low dose of docetaxel plus a standard dose of cisplatin for patients with metastatic non-small-cell lung cancer (NSCLC).
Methods. Eligibility criteria included metastatic disease (stage IV) of NSCLC and a performance status (PS) of 0-2. Cisplatin 80 mg/m2 was given i.v. on day 1 and docetaxel 60 mg/m2 was given i.v. on day 1. Treatment was repeated every 3 to 4 weeks.
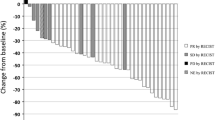
Results. Forty-five patients were enrolled in the study, and the median age was 63 years. Forty-two patients (93%) had a PS of 0-1 and 38 (84%) received two to four courses of chemotherapy. The principal toxicity was neutropenia, and grade 3/4 occurred in 36%/49%. Other hematologic toxicities were mild. Of the 45 patients, subsequent chemotherapy was delayed due to toxicities in only 5 patients (11%), and dose modifications were needed in only 3 patients (7%). There were no treatment-related deaths. Non-hematological toxicities were relatively mild. Allergy (2%), skin rash (11%), edema (9%), and neuropathy (9%) occurred infrequently, and all were grade 1 toxicity. Of the 45 patients, 19 showed partial response, giving a response rate of 42%. The median survival time was 43.3 weeks, and the 1-year survival rate of all patients was 38.7%.
Conclusion. This cisplatin/docetaxel combination chemotherapy is an active and non-toxic regimen in patients with metastatic NSCLC, a result which suggests that the combination may be suitable for randomized controlled trials.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: February 24, 2000 / Accepted: June 2, 2000
About this article
Cite this article
Okamoto, H., Watanabe, K., Segawa, Y. et al. Phase II study of docetaxel and cisplatin in patients with previously untreated metastatic non-small-cell lung cancer. Int J Clin Oncol 5, 316–322 (2000). https://doi.org/10.1007/PL00012056
Issue Date:
DOI: https://doi.org/10.1007/PL00012056