Summary.
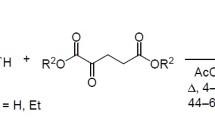
The conjugate addition of malononitrile to α,β-unsaturated ketones catalyzed by piperidine yielded 2-amino-4-aryl-6-methyl-5-(phenylthio)-4H-pyran-3-carbonitriles. The reaction of α,β-unsaturated ketones with cyanoacetamide led to 2-pyridone derivatives. The reaction of malononitrile and ethyl cyanoacetate with an enaminone occurred under elimination of dimethylamine to yield 2-pyridone.
Zusammenfassung.
Die konjugierte Addition von Malonitril an α,β-ungesättigte Ketone, die durch Piperidin katalysiert wird, ergibt 2-Amino-4-aryl-6-methyl-5-(phenylthio)-4H-pyran-3-carbonitrile. Die Reaktion α,β-ungesättigter Ketone mit Cyanacetamid führt zu Pyridonderivaten. Die Umsetzung von Malonitril und Ethylcyanoacetat mit einem Enaminon erfolgt unter Eliminierung von Dimethylamin und liefert 2-Pyridonderivate.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received June 29, 1998. Accepted (revised) August 24, 1998
Rights and permissions
About this article
Cite this article
Bogdanowicz-Szwed, K., Budzowski, A. Synthesis of Phenylthio Substituted 4H-Pyrans and 2-Pyridinones by Conjugate Addition-Cyclization of CH-Acids to α,β-Unsaturated Ketones. Monatshefte fuer Chemie 130, 545–554 (1999). https://doi.org/10.1007/PL00010233
Published:
Issue Date:
DOI: https://doi.org/10.1007/PL00010233