Summary.
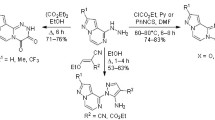
Several pyrazino[2,3-b]phenazine derivatives were prepared by the reaction of 2,3-diaminophenazine with different 1,2-diketones. Nucleophilic substitution of 2,3-dibromomethyl-pyrazino[2,3-b]phenazine with propanol, morpholine, and potassium thiocyanate gave 2,3-bis-(propoxymethyl)-pyrazino[2,3-b]phenazine, 2,3-bis-(4-morpholinylmethyl)-pyrazino[2,3-b]phenazine, and 2,3-bis-(cyanosulfanylmethyl)-pyrazino[2,3-b]phenazine. 2-Aryl-1H-imidazo[4,5-b]phenazine derivatives were synthesized by a one-pot reaction of 2,3-diaminophenazine with different aromatic aldehydes or acids. Reaction of 2,3-diaminophenazine with acetic anhydride and formic acid afforded 1H-imidazo[4,5-b]phenazine and 2-methyl-1H-imidazo[4,5-b]phenazine. Chemical and spectroscopic evidences for the product structures of the new compounds are presented.
Zusammenfassung.
Einige Pyrazino[2,3-b]phenazinderivate wurden durch Reaktion von 2,3-Diaminophenazin mit verschiedenen 1,2-Diketonen dargestellt. Nucleophile Substitution von 2,3-Dibrommethyl-pyrazino[2,3-b]phenazin mit Propanol, Morpholin und Kaliumthiocyanat ergab 2,3-Bis(propoxymethyl)-pyrazino[2,3-b]phenazin, 2,3-Bis-(4-morpholinylmethyl)-pyrazino[2,3-b]phenazin und 2,3-Bis-(cyanosulfanylmethyl)-pyrazino[2,3-b]phenazin. 2-Aryl-1H-imidazo[4,5-b]phenazinderivate wurden in einer Eintopfreaktion aus 2,3-Diaminophenazin mit verschiedenen aromatischen Aldehyden oder Carbonsäuren hergestellt. Reaktion von 2,3-Diaminophenazin mit Essigsäureanhydrid oder Ameisensäure lieferte 1H-Imidazo[4,5-b]phenazin und 2-Methyl-1H-imidazo[4,5-b]phenazin. Chemische und spektroskopische Nachweise für die Struktruren der neuen Verbindungen werden mitgeteilt.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received December 29, 1998. Accepted February 2, 1999
Rights and permissions
About this article
Cite this article
Amer, A., El-Bahnasawi, A., Mahran, M. et al. On the Synthesis of Pyrazino[2,3-b]phenazine and 1H-Imidazo[4,5-b]phenazine Derivatives. Monatshefte fuer Chemie 130, 1217–1225 (1999). https://doi.org/10.1007/PL00010183
Published:
Issue Date:
DOI: https://doi.org/10.1007/PL00010183