Abstract
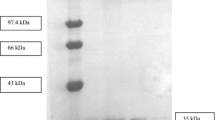
A lipase from a wild strain of Penicillium citrinum was purified by ammonium sulphate precipitation, gel filtration chromatography on a Superose 6 column and hydrophobic interaction chromatography (HIC) on a Phenyl Superose column. The yield and purification factor were 15.2% and 379 fold, respectively. The gel filtration step was efficiently scaled-up in a Superose 6 preparative grade column and after this step, the lipase was recovered in the form of a high molecular weight aggregate. The partial disaggregation of the complex was achieved by HIC and elution with 1.0% (w/v) CHAPS. The lipase produced by Penicillium citrinum forms a dimmer of 63 000 Da, as determined by SDS-PAGE, and it accumulates in the fermentation broth as high molecular weight aggregates (>2 00 000 Da). The analysis of the dimmer showed two subunits with similar molecular weights (31 000–33 000 Da) and isoelectric points (4.8–5.0).
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 22 January 1998
Rights and permissions
About this article
Cite this article
Krieger, N., Taipa, M., Melo, E. et al. Purification of a Penicillium citrinum lipase by chromatographic processes. Bioprocess Engineering 20, 59–65 (1999). https://doi.org/10.1007/PL00009034
Issue Date:
DOI: https://doi.org/10.1007/PL00009034