Abstract
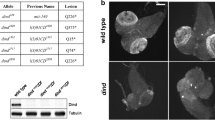
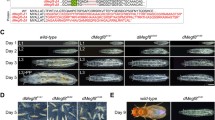
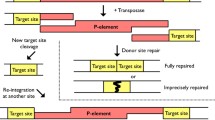
The Ketel gene of Drosophila melanogaster was identified by four KetelD dominant female-sterile mutations and their 27 revertants. The X-ray and the P-induced KetelRalleles delineated the Ketel locus to the 38E1.2–3 cytological position. Although oogenesis proceeds normally in the KetelD/+ females, embryogenesis comes to a deadlock shortly after fertilization inside the normal-looking eggs of the KetelD/+ females. The KetelD alleles are dominant negative mutations of antimorph type. Cytoplasm of the KetelD/+-derived eggs induce lesions when injected into wild-type eggs and the KetelD alleles can be reverted. Zygotes homozygous for loss-of-function (revertant) KetelRalleles die in second larval instar. Analysis of the cold-sensitive Ketel alleles and the genetic interactions between importin-α and KetelR mutant alleles indicate an involvement of the Ketel gene product in oo-, embryogenesis and larval life and show interaction of the KETEL protein with different components of nuclear processes. Molecular analysis (to be published elsewhere) confirmed the genetic data and revealed that the Ketel gene encodes the Orosophila homologue of importin-β, an essential component of nuclear protein import.
Similar content being viewed by others
References
Anderson, K. (1995) One signal, two body axes. Science 269, 489–490.
Choller, R., Anderson, L. L. (1977) Conformational changes associated with the reversible cold inactivation of ribulose-l,5-bisphosphate carboxylase-oxygenase. Biochim. Biophys. Acta 482, 228–240.
Dasso, M., Seki, T., Azuma, Y., Ohba, T., Nishimoto, T. (1994) A mutant form of the Ran/TC4 protein disrupts nuclear function in Xenopus laevis egg extracts by inhibiting the RCC1 protein, a regulator of chromosome condensation. EMBO J. 13, 5732–5744.
Demeter, J., Morphew, M., Sazer, S. (1995) A mutation in the RCCl-related protein piml results in nuclear envelope fragmentation in fission yeast. Proc. Natl. Acad. Sci. USA 28, 1436–1440.
Erdélyi, M., Szabad, J. (1989) Isolation and characterization of dominant female sterile mutations of Drosophila melanogaster. I. Mutations on the third chromosome. Genetics 122, 111–127.
Foe, V. E., Odell, G. M., Edgar, B. A. (1993) Mitosis and morphogenesis in the Drosophila embryo: point and counterpoint. In: Bate, M., Martinez-Arias, A. (eds) The Development of Drosophila melanogaster. Cold Spring Harbor Laboratory Press, New York, pp. 149–300.
Görlich, D., Mattaj, I. W. (1996) Nucleocytoplasmic transport. Science 271, 1513–1518.
Görlich, D., Prehn, S., Laskey, R. A., Hartmann, E. (1994) Isolation of protein that is essential from the first step of nuclear protein import. Cell 79, 767–778.
González-Reyes, A., Elliot, H., St. Johnston, D. (1995) Polarization of both major body axes in Drosophila by gurken-torpedo signalling. Nature 375, 654–658.
Jürgens, G. (1987) Segmental organisation of the tail region in the embryo of Drosophila melanogaster. A blastoderm fate map of the cuticle structures of the larval tail region. Roux’s Arch. Dev. Biol. 196, 141–157.
Jürgens, G., Lehmann, R., Schardin, M., Nüsslein-Volhard, C. (1986) Segmental organisation of the head in the embryo of Drosophila melanogaster. Roux’s Arch. Dev. Biol. 195, 359–377.
Kornblut, S., Dasso, M., Newport, J. (1994) Evidence for a dual role for TC4 protein in regulating nuclear structure and cell cycle progression. J. Cell Biol. 125, 705–719.
Kussel, P., Frasch, M. (1995) Pendulin, a Drosophila protein with cell cycle-dependent nuclear localization, is required for normal cell proliferation. J. Cell Sci. 129, 1491–1507.
Lindsley, D. L., Zimm, G. G. (1992) The Genome of Drosophila melanogaster. Academic Press, San Diego and London.
Lippai, M., Mihály, J., Erdélyi, M., Máthé, E., Puro, J., Belecz, I., Tirian, L., Pósfai, J., Udvardy, A., Boros, I., Szabad, J. (1987) The Ketel gene encodes the Drosophila homologue of importin 90. (Submitted for publication.)
Montell, D. J., Keshishian, H., Spradling, A. C. (1991) Laser ablation studies of the role of the Drosophila oocyte nucleus in pattern formation. Science 254, 290–293.
Panté, N., Aebi, U. (1996) Toward molecular dissection of protein import into nuclei. Current Opinion in Cell Biology 8, 397–406.
Perrimon, N., Mahowald, A. P. (1986) Maternal role of zygotic lethals during early embryogenesis in Drosophila. In: Gametogenesis and the Early Embryo. A. R. Liss Inc., New York, pp. 221–235.
Powers, M. A., Forbes, D. J. (1984) Cytosolic factors in nuclear transport: what is importin? Cell 79, 931–934.
Rexach, M., Blobel, G. (1995) Protein import into nuclei: association and dissociation reactions involving transport substrate, transport factors and nucleoporins. Cell 83, 683–692.
Schlenstedt, G. (1996) Protein import into the nucleus. FEBS Letters 389, 75–79.
Schüpbach, T. (1987) Germ line and soma cooperate during oogenesis to establish the dorsoventral pattern of egg shell and embryo in Drosophila melanogaster. Cell 49, 699–707.
Schüpbach, T., Roth, S. (1994) Dorsoventral patterning in Drosophila oogenesis. Current Biol. 4, 502–507.
Schüpbach, T., Wieschaus, E. (1989) Female sterile mutations on the second chromosome of Drosophila melanogaster. 1. Maternal effect mutations. Genetics 121, 101–117.
Szabad, J., Erdélyi, M., Hoffmann, G., Szidonya, J., Wright, T. R. F. (1989) Isolation and characterization of dominant female sterile mutations of Drosophila melanogaster. II. Mutations on the second chromosome. Genetics 122, 823–835.
Török, I., Strand, D., Schmitt, R., Tick, G., Török, T., Kiss, I., Mechler, B. M. (1995) The overgrown hematopoietic organs-31 tumor suppressor gene of Drosophila encodes an importin-like protein accumulating in the nucleus at the onset of mitosis. J. Cell Biol. 129, 1473–1489.
Tricoire, H. (1988) Dominant maternal interactions with Drosophila segmentation genes. Wilhelm Roux’s Arch. Dev. Biol. 197, 115–123.
Wieschaus, E. (1996) Embryonic transcription and the control of developmental pathways. Genetics 142, 5–10.
Wieschaus, E., Nüsslein-Volhard, C. (1986) Looking at embryos. In: Roberts, D. B. (ed.) Drosophila a Practical Approach. I.R.L. Press, Oxford, pp. 199–227.
Author information
Authors and Affiliations
Additional information
Dedicated to the memory of Professor Gábor Szabó.
Rights and permissions
About this article
Cite this article
Erdélyi, M., Máthé, E. & Szabad, J. Genetic and developmental analysis of mutant Ketel alleles that identify the Drosophila importin-β homologue. BIOLOGIA FUTURA 48, 323–338 (1997). https://doi.org/10.1007/BF03543204
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03543204