Abstract
Background
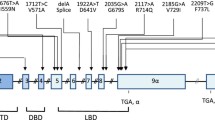
Alternative splicing of the transcripts of the human glucocorticoid receptor gene results in two mutually exclusive products, the classic, ligand-binding glucocorticoid receptor (hGRα), and a dominant negative non-ligand-binding isoform, hGRβ.
Materials and Methods
We examined the existence of and quantified both hGRα and hGRβ isoforms in a panel of human tissues, as well as in intact and fractionated HeLa cells, using specific quantitative Western blots and/or immunocytochemistry. We studied the potential interactions of hGRβ with heat shock protein (hsp) 90 and/or hGRα using cross-immunoadsorption/precipitation procedures followed by Western blots.
Results
For the first time, we demonstrated the natural existence of the hGRβ protein, which was widely expressed in human tissues. The ratio of immunoreactive hGRα to hGRβ varied from 0.2 to 1.0 among different tissues, and was approximately 0.2 in HeLa cells. In the latter, both isoforms were distributed in the cytoplasm and nucleus in the absence of the hormonal ligand, and translocated into the nucleus after addition of dexamethasone. The cytosolic and nuclear hGRα-to-hGRβ ratio remained the same before and after dexamethasone exposure, suggesting that upon activation the two isoforms translocated into the nucleus in equal proportions. hGRα- and hGRβ-specific antibodies cross-adsorbed and precipitated cytosolic and nuclear glucocorticoid hGRα and hGRβ, respectively, as well as hsp90, suggesting that hGRα and hGRβ are in complex with hsp90 and/or each other.
Conclusions
The hGRβ protein is widely expressed throughout the human body and present mostly in the cytoplasm of human cells, in complex with hsp90 and other proteins. In the presence of glucocorticoid, hGRβ probably heterodimerizes with ligand-bound hGRα and translocates into the nucleus to act as a dominant negative inhibitor of the classic receptor.






Similar content being viewed by others
References
Hollenberg SM, Weinberger C, Ong ES, et al. (1985) Primary structure and expression of functional human glucocorticoid receptor cDNA. Nature 318: 635–641.
Encio IJ, Detera-Wadleigh SD. (1991) The genomic structure of the human glucocorticoid receptor. J. Biol. Chem. 266: 7182–7188.
Giguere V, Hollenberg SM, Rosenfeld MG, Evans RM. (1986) Functional domains of human glucocorticoid receptor. Cell 46: 645–652.
Hurley DM, Accili D, Stratakis CA, et al. (1991) Point mutation causing a single amino acid substitution in the hormonebinding domain of the glucocorticoid receptor in familial glucocorticoid resistance. J. Clin. Invest. 87: 680–686.
Karl M, Lamberts SWJ, Detera-Wadleigh SD, et al. (1993) Familial glucocorticoid resistance caused by a splice site deletion in the human glucocorticoid receptor gene. J. Clin. Endocrinol. Metab. 76: 683–689.
Chrousos GP, Detera-Wadleigh SD, Karl M. (1993) Syndromes of glucocorticoid resistance. Ann. Intern. Med. 119: 1113–1124.
Bamberger CM, Bamberger A-M, Castro M, Chrousos GP. (1995) Glucocorticoid receptor-β, a potential endogenous inhibitor of glucocorticoid action in humans. J. Clin. Invest. 95: 2435–2441.
Oakley RH, Sar M, Cidlowski JA. (1996) The human glucocorticoid receptor β isoform. J. Biol. Chem. 271: 9550–9559.
Yamamoto KR. (1985) Steroid receptor regulated transcription of specific genes and gene networks. Annu. Rev. Genet. 19: 209–252.
Picard D, Yamamoto KR. (1987) Two signals mediate hormone-dependent nuclear localization of the glucocorticoid receptor. EMBO J. 6: 3333–3340.
Evans RM. (1988) The steroid and thyroid hormone receptor superfamily. Science 240: 889–895.
Picard D, Salserand JS, Yamamoto KR. (1988) A movable and regulable inactivation function within the steroid binding domain of the glucocorticoid receptor. Cell 54: 1073–1080.
Truss M, Beato M. (1993) Steroid hormone receptors: Interaction with deoxyribonucleic acid and transcription factors. Endocr. Rev. 14: 459–479.
Kaumaya PTP, Berndt KDA, Heidorn DB, Ttrewhella J, Kezdy FJ, Goldberg E. (1990) Synthesis and biophysical characterization of engineered topograph immunogenic determinant with α α topology. Biochemistry 29: 13–23.
Ulrich SM, Moore SK, Appella E, May P. (1989) Transcriptional and translation analysis of the murine 84- and 86-KDa heat shock proteins. J. Biol. Chem. 264: 6810–6816.
Laemmli UK. (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685.
Elliot S, Goldsmith P, Knepper M, Haughey M, Olson B. (1996) Urinary excretion of aquaporin-2 in humans: a potential marker of collecting duct responsiveness to vasopressin. J. Am. Soc. Nephrol. 7: 403–409.
Tomita M, Chrousos GP, Brandon DD, et al. (1985) Glucocorticoid receptors in Epstein-Barr virus-transformed human lymphocytes. Horm. Metabol. Res. 17: 674–678.
Karl M, Steven SWJ, Koper JW, et al. (in press) Cushing’s disease preceded by generalized glucocorticoid resistance: clinical consequences of a novel, dominant-negative glucocorticoid receptor mutation. PAAP. 108: 296–307.
Wang Y, Micsicek RJ. (1991) Identification of a dominant negative form of the human estrogen receptor. Mol. Endocrinol. 5: 1707–1715.
Vegeto E, Shahbaz MM, Wen DX, Goldman ME, O’Malley BW. (1993) Human progesterone A form is a cell- and promoter-specific repressor of human progesterone receptor B function. Mol. Endocrinol. 7: 1244–1255.
Katz D, Lazar MA. (1993) Dominant negative activity of an endogenous thyroid hormone receptor variant (α2) is due to competition for binding sites on target genes. J. Biol. Chem. 268: 20904–20910.
Nagaya T, Jameson JL. (1993) Thyroid hormone resistance syndrome: Correlation of dominant negative activity and location of mutations. J. Clin. Endocrinol. Metab. 77: 982–990.
Cooney AJ, Leng X, Tsai Si, Tsai M-J, O’Malley BW. (1993) Multiple mechanism of chicken ovalbumin upstream promotor transcription factor-dependent repression of transactivation by the vitamin D, thyroid hormone, and the retinoic acid receptors. J. Biol. Chem. 268: 4252–4160.
Liu R-T, Suzuki S, Miyamoto T, Takeda T, Ozata M, DeGroot LJ. (1995) The dominant negative effect of thyroid receptor splicing variant α2 does not require binding to a thyroid response element. Mol. Endocrinol. 9: 86–95.
Pratt WB. (1993) The role of heat shock protein in regulating the function, folding, and trafficking of the glucocorticoid receptor. J. Biol. Chem. 268: 21455–21458.
Pratt WB, Sanches ER, Bresnick EH, et al. (1989) Interaction of the glucocorticoid receptor with the MW 90,000 heat shock protein: An envolving model of ligand-mediated transformation and translocation. Cancer Res. 49: 2222–2229.
Dalman CF, Scherrer CL, Taylors PL, Akil H, Pratt WB. (1991) Localization of the 90-kDa heat shock protein-binding site within the hormone-binding domain of the glucocorticoid receptor by peptide competition. J. Biol. Chem. 266: 3482–3490.
Tsai M-J, O’Malley BW. (1994) Molecular mechanism of action of steroid/thyroid receptor superfamily members. Annu. Rev. Biochem. 63: 451–479.
Smith DF. (1993) Dynamics of heat shock protein 90-progesterone receptor binding and the disactivation loop model for steroid receptor complexes. Mol. Endocrinol. 7: 1418–1429.
Tilders FJ, DeRijk RH, Van Damm AM, Vincent VA, Schotanus K, Persoons JH. (1994) Activation of the hypothalamic-pituitary-adrenal axis by bacterial endotoxins: Routes and intermediate signals. Psychoneuroendocrinology 19: 209–232.
Chrousos GP. (1995) The hypothalamic-pituitary-adrenal axis and immune-mediated inflammation. N. Engl. J. Med. 332: 1351–1362.
Chrousos GP, Castro M, Leung DY, et al. (in press) Molecular mechanisms of glucocorticoid resistance/hypersensitivity: Potential clinical implications. Am. J. Respir. Crit. Care Med.
Sher ER, Leung DY, Surs W, et al. (1994) Steroid-resistant asthma. Cellular mechanisms contributing to inadequate responses to glucocorticoid therapy. J. Clin. Invest. 93: 33–39.
Castro M, Leung DY, Karl M, et al. (1996) Changes in the alternative splicing of the glucocorticoid receptor gene might contribute to the pathogenesis of glucocorticoid-resistant asthma. Program of the 10th International Congress of Endocrinology, San Francisco. Abstract.
Acknowledgments
We thank Keith Zachman for superb technical assistance and Dr. B. K. Kishore, NHLBI, for his assistance with the quantitative Western blots. M. de Castro is supported by Fundacao de Amparo a Pesquisa do Estado de Sao Paulo (FAPESP—Proc 93/3735-6), Brasil.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
de Castro, M., Elliot, S., Kino, T. et al. The Non-Ligand Binding β-Isoform of the Human Glucocorticoid Receptor (hGRβ): Tissue Levels, Mechanism of Action, and Potential Physiologic Role. Mol Med 2, 597–607 (1996). https://doi.org/10.1007/BF03401643
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03401643