Abstract
OBJECTIVE
To compare the thyroid hormonal responses to high-intensity interval exercise (IE) and steady-state endurance exercise (SEE) in highly trained males (n=15).
DESIGN
The IE session consisted of repeated periods of 90-seconds treadmill running at 100–110% VO2max and 90-seconds active recovery at 40% VO2max for 42–47 minutes. The SEE session was a 45-minute run at 60–65% VO2max. Total work output was equal for each session. A 45-minute supine rest control session (CON) was also performed. Pre-session (PRE), immediate post-session (POST), and 12-hours post-session (12POST) blood samples were collected and used to determine free (f) T4, fT3, reverse (r) T3, and cortisol levels.
RESULTS
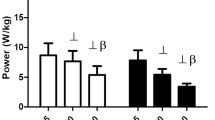
All PRE hormone levels were within clinical norms and did not differ significantly between sessions. All POST IE and SEE hormone levels were significantly elevated compared to POST CON (p<0.001). At 12POST, no significant differences between CON and SEE hormonal levels were observed; however, fT3 was significantly reduced and rT3 was significantly elevated in 12POST IE compared to 12POST SEE and CON (p=0.022). For IE, at 12POST a negative correlation (rs = -0.70, p<0.004) was found between fT3 and rT3. Also, for IE, a positive correlation (rs = 0.74, p<0.002) between cortisol POST and rT3 12POST was noted, and a negative correlation (rs = -0.72, p<0.003) between cortisol POST and fT3 12POST.
CONCLUSION
IE results in a suppressed peripheral conversion of T4 to T3 implying that a longer recovery period is necessary for hormonal levels to return to normal following IE compared to SEE. These findings are useful in the implementation of training regimens relative to recovery needs and prevention of over-reaching -overtraining.
Similar content being viewed by others
References
Moore AW, Timmerman S, Brownlee KK, Rubin DA, Hackney AC, 2005 Strenuous, fatiguing exercise: relationship of cortisol to circulating thyroid hormones. Int J Endocrinol Metab 1: 18–24.
Aakvaag A, Sand T, Opstad PK, Fonnum F, 1978 Hormonal changes in serum in young men during prolonged physical strain. Eur J Appl Physiol Occup Physiol 39: 283–291.
Brooks GA, Fahey TD, White TP 1996 Neuralendocrine control of metabolism. In: Exercise physiology: human bioenergetics and its application, 2nd edition, Mayfield Publishing, Toronto; pp, 56–196.
Hackney AC 2011 Thyroid Axis, Prolactin and Exercise. In: Ghigo E, Lanfranco F, Strasburger CJ (eds), Endocrine updates: hormone use and abuse by athletes, Springer-Verlag Publisher, Stuttgart; Vol 29, pp, 17–24.
Ciloglu F, Peker I, Pehlivan A, et al, 2005 Exercise intensity and its effects on thyroid hormones. Neuro Endocrinol Lett 26: 830–834.
Joyner MJ, Coyle EF, 2008 Endurance exercise performance: the physiology of champions. J Physiol 586: 35–44.
Laursen PB, Jenkins DG, 2002 The scientific basis of high intensity interval training. Sports Med 32: 53–73.
McMurray RG, Hackney AC 2000 The endocrine system and exercise. In: Exercise & sports science. Garrett W & Kirkendal LD (eds), Williams & Wilkins Publisher, Philadelphia; pp, 135–162.
Jackson AS, Pollock ML, 1978 Generalized equations for predicting body density of men. Brit J Nutr 40: 497–504.
American College of Sports Medicine 2009 Guidelines for exercise testing and prescription, Lippincott, Williams & Wilkins, Philadelphia; pp, 68–75.
Daly W, Seegers CA, Rubin DA, Dobridge JD, Hackney AC, 2005 Relationship between stress hormones and testosterone with prolonged endurance exercise. Eur J Appl Physiol 93: 375–380.
Dill DB, Costill DL, 1974 Calculation and percentages in volumes of blood, plasma, and red cells in dehydration. J Appl Physiol 37: 247–248.
Hackney AC, Viru A, 2008 Research methodology: issues with endocrinological measurements in exercise science and sport medicine. J Athletic Training 43: 631–639.
Levene H 1960 Robust Tests for Equality of Variances. In: Contributions to Probability and Statistics, Olkin I (ed), Stanford University Press, Palo Alto, CA; pp, 278–292.
Greenhouse SW, Geisser S, 1959 On methods in the analysis of profile data. Psychometrika 24: 95–112.
Tietz NW 1990 Clinical Guide to Laboratory Tests Saunders Publishing, Philadelphia; pp. 78–130.
Viru A, Viru M 2001 Biochemical monitoring of sport training. Human Kinetics Publishing, Champaign IL; pp, 91–142.
Galbo H 1983 Hormonal and metabolic adaptation to exercise. Georg Thieme Verlag, Stuttgart; pp, 21–110.
Kuoppasalmi K, Näveri H, Rehunen S, Härkönen M, Adlercreutz H, 1976 Effect of strenuous anaerobic running exercise on plasma growth hormone, cortisol, luteinizing hormone, testosterone, androstenedione, estrone and estradiol. J Steroid Biochem 7: 823–829.
Opstad PK, Aakvaag A, 1981 The effect of a high calorie diet on hormonal changes in young men during prolonged physical strain and sleep deprivation. Eur J Appl Physiol Occup Physiol 46: 31–39.
Hackney AC, Hodgdon JA, Hesslink R, Trygg K, 1995 Thyroid hormone responses to military winter activities in the Arctic Region. Arctic Medical Res 54: 82–90.
Lehmann M, Knizia K, Gastmann U, et al, 1993 Influence of 6-week, 6 days per week, training on pituitary function in recreational athletes. Brit J Sports Med 27: 186–192.
Lehmann M, Lormes W, Opitz-Gress A, et al, 1997 Training and overtraining: an overview and experimental results in endurance sports. J Sports Med Physical Fit 37: 7–17.
Urhausen A, Gabriel H, Kindermann W, 1995 Blood hormones as markers of training stress and overtraining. Sports Med 20: 251–276.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hackney, A.C., Kallman, A., Hosick, K.P. et al. Thyroid hormonal responses to intensive interval versus steady-state endurance exercise sessions. Hormones 11, 54–60 (2012). https://doi.org/10.1007/BF03401537
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03401537