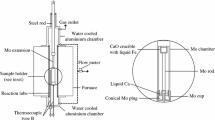
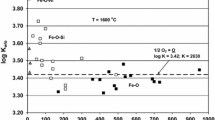
Abstract
The solubility of oxygen in iron containing aluminum has been determined at 1550°, 1600°, and 1650°C and found to be much higher than predicted from theoretical considerations, possibly due to equilibria with an iron-aluminum spinel phase rather than pure Al2O3. The presence of manganese greatly increased the deoxidizing power of aluminum.
Similar content being viewed by others
References
“Basic Open Hearth Steelmaking.” Physical Chemistry of Steelmaking Committee, AIME, New York, N. Y., 1944.
C. Benedicks and H. Löfquist: Nonmetallic Inclusions in Iron and Steel. John Wiley and Sons, Inc., New York, N. Y., 1931.
F. Körber and W. Oelsen: Über die Beziehungen zwischen manganhaltigem Eisen and Schlacken, die fast nur aus Manganoxydul and Eisenoxydul besteben. Mitt. K.-W. Inst. f.d. Eisenforsch., Dusseldorf, (1932) 14, 181–204.
F. Körber and W. Oelsen: Die Grundlagen der Desoxydation mit Mangan und Silizium. Mitt. K.-W. Inst. f.d. Eisenforsch., Dusseldorf, (1933) 15, 271–309.
C. H. Herty, Jr. and G. R. Fitterer: The Physical Chemistry of Steelmaking: Deoxidation with Silicon and the Formation of Ferrous Silicate Inclusions in Steel. U. S. Bur. of Mines, Carnegie Inst. of Tech., and Min. and Met. Advisory Boards Coop. Bull. 36, Pittsburgh, Pa. 1928.
C. H. Herty, Jr., G. R. Fitterer, and J. M. Byrns: The Physical Chemistry of Steelmaking: Deoxidation of Steel with Aluminum. Ibid., Bull. 46.
W. Krings and H. Schackman: Über Gleichgewichte zwischen Metallen and Schlacken im Schmelzflusse, I. Ztsch. anorg. allg. Chem. (1931) 202, 99–112.
H. Wentrup and G. Hieber: Umsetzungen zwischen Aluminum and Sauerstoff in Eisenschmelzen. Archiv. f. d. Eisenhuttenw. (1939) 13, 15–20.
H. Schenck and E. O. Brüggeman: Untersuchungen über die Chemie des sauren Siemens-Martin-Verfahrens. Archiv. f. d. Eisenhuttenw. (1935–1936) 9, 543–553.
H. C. Vacher and E. H. Hamilton: The Carbon-oxygen Equilibrium in Liquid Iron. Trans. AIME (1931) 95, 124–140.
J. Chipman: Deoxidation of Steel. Trans. A.S.M. (1934) 22, 385–433.
S. Marshall and J. Chipman: The Carbon-oxygen Equilibrium in Liquid Iron. Trans. A.S.M. (1942) 30, 695–746.
J. Chipman and N. J. Grant: The Induction Furnace as a High-Temperature Calorimeter and the Heat of Solution of Silicon in Liquid Iron. Trans. A.S.M. (1943) 31 365–379.
D. C. Hilty and Walter Crafts: The Solubility of Oxygen in Liquid Iron Containing Silicon and Manganese. Trans. AIME (1950) 188, 425. Jnl. of Metals, Feb. 1950.
C. R. Taylor and J. Chipman: Equilibria of Liquid Iron and Simple Basic and Acid Slags in a Rotating Induction Furnace. Trans. AIME (1943) 154, 228–245.
E. P. Barrett, W. F. Holbrook, and C. E. Wood: Induction Furnaces for Rotating Liquid Crucibles. Trans. AIME (1939) 135, 73–84.
W. A. Hammond and J. R. Withrow: Soluble Anhydrite as a Dessicating Agent. Ind. and Eng. Chem. (1935) 25, 653–659.
British Iron and Steel Research Association: Symp. on the Contamination of Platinum Thermocouples. Jnl. Iron and Steel Inst., London, (1947) 155, 213–231.
C. E. Sims and G. A. Lillieqvist: Inclusions — Their Effect, Solubility, and Control in Cast Steel. Trans. AIME (1932) 100, 154–195.
W. Crafts, J. J. Egan, and W. D. Forgeng: Formation of Inclusions in Steel Castings. Trans. AIME, (1940) 140, 233–262.
J. White: The Physical Chemistry of Open-hearth Slags. Jnl. Iron and Steel Inst. London, (1943) 148, II 579–688.
R. Schenck, H. Franz, and H. Willeke: Gleichgewichtsuntersuchungen über die Reduktions — Oxydations — und Kohlungsvorgange beim Eisen, IX., Ztsch. f. anorg u. allge. Chemie (1929) 184, 1–38.
J. Chipman: Another Look at the Problem of Steel Deoxidation. Metal Progress, (1949) 56, No. 2, 211–221.
Author information
Authors and Affiliations
Additional information
AIME New York Meeting, Feb. 1950.
TP 2805 C. Discussion (2 copies) may be sent to Transactions AIME before Apr. 1, 1950, and will be published Nov. 1950.
Rights and permissions
About this article
Cite this article
Hilty, D.C., Crafts, W. The solubility of oxygen in liquid iron containing aluminum. JOM 2, 414–424 (1950). https://doi.org/10.1007/BF03399019
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03399019