Summary
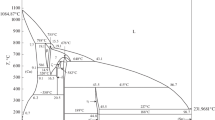
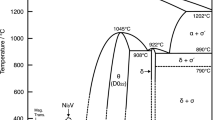
1—Diffusion coefficients in the Cu-Pd system have been determined as a function of temperature and concentration. The concentration dependence is slight in both extremes of concentration and strong at intermediate concentrations. The diffusion coefficient approaches the value of the self-diffusion coefficient of copper in dilute palladium solution.
2—A redetermination of the dependence of the diffusion coefficients at the high copper end of the Cu-Ni system confirmed a strong dependence of the diffusion coefficient on composition in the relatively dilute solutions.
3—Individual diffusion coefficients for copper and nickel atoms have been found by the use of markers at 89.9 atomic pct Cu. DCu is about 30 pet greater than DNi over the temperature range 923° to 1049°C.
4—The deviation of the diffusion coefficient at infinite dilution from the self-diffusion coefficient of the solvent metal is generally greater the greater the melting-point depression. Both factors also tend to become greater the more limited the solid solubility of the solute metal.
Similar content being viewed by others
References
L. Boltzmann: Annalen Physik (1894) 53, p. 960.
C. Matano: Journal of Physics (1933) 8, p. 109.
G. Grube and A. Jedele: Ztsch Elektrochemie (1932) 38, p. 799.
L. Onsager: Physical Review (1931) 37 p 405
J. C. Fisher, J. H. Hollomon, and D. Turnbull. Trans. AIME (1948) 175, p. 202; Metals Technology (February 1948).
B. V. Rollin: Physical Review (1939) 55, p. 231.
J. Steigman, W. Shockley, and F. C. Nix: Physical Review (1939) 56, p. 13.
C. L. Raynor, L. Thomassen, and L. J. Rouse: Trans. ASM (1942) 30, p. 3.
M. S. Maier and H. R. Nelson: Trans. AIME (1942) 147 p. 39.
C. Wells and R. F. Mehl: Trans. AIME (1941) 145, p. 329.
L. C. C. da Silva and R. F. Mehl: Trans. AIME (1951) 191, p. 155; Journal of Metals (February 1951).
W. A. Johnson: Trans. AIME (1942) 147, p. 331.
F. N. Rhines and R. F. Mehl: Trans. AIME (1938) 128, p. 185.
L. S. Darken: Trans. AIME (1948) 175, p. 184; Metals Technology (January 1948).
R. F. Mehl, F. N. Rhines, and K. A. von Steinen: Metals and Alloys (1941) 13, p. 41.
W. Seith: Ztsch. Elektrochemie (1935) 41, p. 872.
C. E. Birchenall and R. F. Mehl: Trans. AIME (1950) 188, p. 144; Journal of Metals (January 1950).
R. M. Barrer: Diffusion In and Through Solids, p. 283 (1941) Cambridge.
L. Claire: Report on Progress in Metal Physics. Edited by Chalmers. (1949) London. Butterworths.
J. C. Slater: Introduction to Chemical Physics, pp. 475 ff. (1939) McGraw-Hill Book Co.
C. Matano: Japanese Journal of Physics (1934) 9, p. 41.
Martin and Asara: Physical Review (1950) 80, p. 123.
R. C. Ruder: Doctorate Thesis, Carnegie Institute of Technology. (1950).
Author information
Authors and Affiliations
Additional information
Discussion on this paper, TP 3329E. may be sent, 2 copies, to AIM. by Dec. 1, 1952. Manuscript, March 12, 1952. Philadelphia Meeting, October 1952.
This paper represents part of a thesis by D. E. Thomas submitted in partial fulfillment of requirements for the degree of Doctor of Science to the Graduate Committee of Carnegie Institute of Technology.
Rights and permissions
About this article
Cite this article
Thomas, D.E., Birchenall, C.E. Concentration Dependence of Diffusion Coefficients In Metallic Solid Solution. JOM 4, 867–873 (1952). https://doi.org/10.1007/BF03398155
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03398155