Abstract
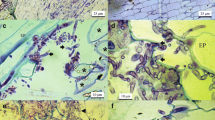
Clavibacter michiganensis subsp. michiganensis (Cmm) causes bacterial canker in tomatoes representing a significant threat to worldwide tomato production. In order to demonstrate potential resistance mechanisms in tomato cultivars differing in susceptibility to the pathogen, inoculations were carried out in the greenhouse followed by histological examinations of infected transverse stem sections using histochemical staining methods. In the greenhouse trials, cultivars ‘Moneymaker’ and ‘Lyconorma’ showed significantly stronger disease symptoms than ‘Irat L3’ and ‘Hawaii 7998’ four weeks after inoculation with Cmm. More occlusions of the xylem vessels, but less tyloses were found in the two susceptible cultivars than in the less susceptible cultivars. Further plant responses to the pathogen like accumulation of phenolic compounds in the xylem parenchyma, xylem lignification or callose deposition were observed in stems of infected plants, but these reactions occurred to a similar extent in all four cultivars. Since enhanced accumulation of phenolic substances, lignin and callose was detected in infected plants of all four cultivars they appear to be components of basal resistance of tomato to Cmm. With immuno-fluorescence microscopy, Cmm was localized on the surface of the seed coat but not in the deeper seed layers or the embryo of a moderately susceptible tomato cultivar. After seed germination, bacteria were directly transmitted to the radicle, hypocotyl and cotyledons of seedlings emerging from infected seeds, suggesting an external rather than a systemic transmission of the bacteria from seed to the plant shoot.
Similar content being viewed by others
References
Agrios GN, 2005. Plant Pathology, 5th Edition. Elsevier LTD, Oxford, 952 pp.
Balaji V & Sessa G, 2008. Activation and manipulation of host responses by a Gram-positive bacterium. Plant Signal Behav 3, 839–841.
Balogun OS & Teraoka T, 2004. Time-course analyses of the accumulation of phenols in tomato seedling infected with potato virus X and tobacco mosaic virus. Biokemistri 16, 112–120.
Beckman CH, 2000. Phenolic-storing cells: keys to programmed cell death and periderm formation in wilt disease resistance and in general defence responses in plants? Physiol Mol Plant Pathol 57, 101–110.
Biggerstaff CM, Gleason ML & Braun EJ, 2000. Refinement of a nondestructive tomato seed assay for Clavibacter michiganensis subsp. michiganensis using seed fiber. Seed Sci Technol 28, 261–269.
De Leon L, Siverio F & Rodriguez A, 2006. Detection of Clavibacter michiganensis subsp. michiganensis in tomato seeds using immunomagnetic separation. J Microbiol Meth 67, 141–149.
EPPO, 2013. Diagnostics: Clavibacter michiganensis subsp. michiganensis. OEPP/EPPO Bulletin 43, 46–67.
Francis DM, Kabelka E, Bell J, Franchino B & St. Clair D, 2001. Resistance to bacterial canker in tomato (Lycopersicon hirsutum LA407) and its progeny derived from crosses to L. esculentum. Plant Dis 85, 1171–1176.
Ftayeh R, 2010. Investigations on the elimination of Clavi-bacter michiganensis subsp. michiganensis, the causal agent of bacterial canker, from tomato cultures by highly sensitive detection methods and effective seed treatments. PhD thesis, Georg-August-University, Göttingen.
Ftayeh R, Tiedemann A v. & Rudolph K, 2011. A new selective medium for isolation of Clavibacter michiganense subsp. michiganense from tomato plants and seed. Phytopathology 101, 1355–1364.
Gartemann K-H, Kirchner O, Engemann J, Gräfen I, Eichenlaub R & Burger A, 2003. Clavibacter michiganensis subsp. michiganensis: first steps in the understanding of virulence of a Gram-positive phytopathogenic bacterium. J Biotechnol 106, 179–191.
Jahr H, Bahro R, Burger A, Ahlemeyer J & Eichenlaub R, 1999. Interactions between Clavibacter michiganensis and its host plants. Environ Microbiol 1, 113–118.
Kabelka E, Franchino B & Francis DM, 2002. Two loci from Lycopersicon hirsutum LA407 confer resistance to strains of Clavibacter michiganensis subsp. michiganensis. Phytopathology 92, 504–10.
Mandal S & Mitra A, 2007. Reinforcement of cell wall in roots of Lycopersicon esculentum through induction of phenolic compounds and lignin by elicitors. Physiol Mol Plant Pathol 71, 201–209.
Pegg GF & Dixon GR, 1969. The reactions of susceptible and resistant tomato cultivars to strains of Verticillium albo-atrum. Ann Appl Biol 63, 389–400.
Poysa V, 1993. Evaluation of tomato breeding lines resistant to bacterial canker. Can J Plant Pathol 15, 301–304.
Sandbrink JM, Ooijen JW, Purimahua CC, Vrielink M, Verkerk R, Zabel P & Lindhout P, 1995. Localization of genes for bacterial canker resistance in Lycopersicon peruvianum using RFLPs. Theor Appl Genet 90, 444–450.
Sinha AK & Wood RKS, 1967. An analysis of responses of resistant and of susceptible tomato plants to Verticillium infection. Ann Appl Biol 59, 143–154.
van den Bulk RW, Jansen J, Lindhout WH & Löffler HJM, 1991. Screening of tomato somaclones for resistance to bacterial canker (Clavibacter michiganensis subsp. michi-ganensis). Plant Breeding 107, 190–196.
van Heusden AW, Koornneef M, Voorrips RE, Brüggemann W, Pet G, Vrielink-van Ginkel R, Chen X & Lindhout P, 1999. Three QTLs from Lycopersicon peruvianum confer a high level of resistance to Clavibacter michiganensis ssp. michi-ganensis. Theor Appl Genet 99, 1068–1074.
van Steekelenburg NAM, 1985. Resistance to Cor ynebacterium michiganense in tomato genotypes. Euphytica 34, 245–250.
Xu X, Miller SA, Baysal-Gurel F, Gartemann K-H, Eichenlaub R & Rajashekara G, 2010. Bioluminescence imaging of Clavibacter michiganensis subsp. michiganensis infection of tomato seeds and plants. Appl Environ Microbiol 76, 3978–3998.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stüwe, B., von Tiedemann, A. Bacterial canker of tomatoes — histological characterization of cultivar resistance and seed transmission. J Plant Dis Prot 120, 194–200 (2013). https://doi.org/10.1007/BF03356474
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03356474