Abstract
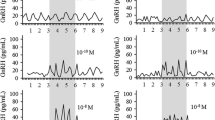
To investigate whether endogenous GABA participates in the control of gonadotropin secretion during the menstrual cycle, placebo or sodium valproate (DPA), an anticonvulsant drug which enhances endogenous GABA content by blocking GABA degradation, were administered to regularly cycling women both during early follicular and midluteal phase. In a first set of experiments, the effect of DPA administration (400 mg, orally) on basal gonadotropin secretion was evaluated in 13 subjects. During early follicular phase (n = 6), no significant changes In plasma gonadotropin levels were observed after DPA or placebo administration. Conversely, during midluteal phase (n = 7), DPA administration resulted in a significant fall (p < 0.01) in plasma LH concentrations, with a maximal percent decrease of 41.8 ± 6.7% after 120 min. No changes in plasma FSH levels were observed. In a second set of experiments, the effect of DPA pretreatment (400 mg, orally) on gonadotropin release stimulated by a pulse of exogenous GnRH (10 μg, iv bolus) was studied in 11 subjects. During both follicular (n = 4) and luteal phase (n = 7), DPA did not modify gonadotropin response to GnRH injected 1h after pretreatment. Finally, 8 subjects were submitted to iv injection with 10 μg GnRH 2h after pretreatment with DPA (400 mg, orally) or placebo. During both follicular (n = 4) and luteal phase (n =4), no statistical differences in gonadotropin response to GnRH were found between DPA and placebo pretreatment. These findings demonstrated that during the estrogen-progesterone (midluteal) phase of menstrual cycle, endogenous GABA is involved in the inhibitory regulation of LH secretion at a central level.
Similar content being viewed by others
References
Racagni G., Apud J.A., Cocchi D., Locatelli V., Müller E.E. GABAergic control of anterior pituitary hormone secretion. Life. Sci. 31: 823, 1982.
Tappaz M.L., Oertel W.H., Wassef M., Mugnaini E. Central GABAergic neuroendocrine regulation: pharmacological and morphological evidence. Prog. Brain Res. 55: 77, 1982.
Simon D., Penry J.K. Sodium di-n-propylacetate (DPA) in the treatment of epilepsy. A review. Epilepsia 16: 549, 1975.
Van der Laan J.W., de Boer T., Bruinvels J. Di-n-propylacetate and GABA degradation. Preferential inhibition of succinic semialdehyde dehydrogenase and indirect inhibition of GABA-transaminase. J. Neurochem. 32: 1769, 1979.
Melis G.B., Paoletti A.M., Mais V., Mastrapasqua N.M., Strigini F., Fruzzetti F., Guarnieri G., Gambacciani M., Fioretti P. The effects of the GABAergic drug, sodium valproate, on prolactin secretion in normal and hyperprolactinemic subjects. J. Clin. Endocrinol. Metab. 54: 485, 1982.
Melis G.B., Fruzzetti F., Paoletti A.M., Mais V., Kemeny A., Strigini F., Boldrini A., Fioretti P. Pharmacological activation of γ-aminobutyric acid-system blunts prolactin response to mechanical breast stimulation in puerperal women. J. Clin. Endocrinol. Metab. 58: 201, 1984.
Melis G.B., Fruzzetti F., Paoletti A.M., Mais V., Beneventi F., Trimarchi G., Fioretti P. Effects of the GABAergic drug, sodium valproate, on the prolactin release evoked by pharmacological stimuli in normal women. Clin. Endocrinol. (Oxf.) 20: 245, 1984.
Ondo J.G. Gamma-aminobutyric acid effects on pituitary gonadotropin secretion. Science 186: 738, 1974.
Pass K.A., Ondo J.G. The effects of γ-aminobutyric acid on prolactin and gonadotropin secretion in the unanesthetized rat. Endocrinology 100: 1437, 1977.
Vijayan E., McCann S.M. The effects of intraventricular injection of γ-aminobutyric acid (GABA) on prolactin and gonadotropin release in conscious female rats. Brain Res. 155: 35, 1978.
Lamberts R., Vijayan E., Graf M., Mansky T., Wuttke W. Involvement of preoptic-anterior hypothalamic GABA neurons in the regulation of pituitary LH and prolactin release. Exp. Brain Res. 52: 356, 1983.
Donoso A.O., Banzan A.M. Effects of increase of brain GABA levels of the hypothalamic-pituitary-luteinizing hormone axis in rats. Acta Endocrinol. (Kbh.) 106: 298, 1984.
Earley C.J., Leonard B.E. GABA and gonadal hormones. Brain Res. 155: 27, 1978.
Mansky T., Mestres-Ventura P., Wuttke W. Involvement of GABA in the feedback action of estradiol on gonadotropin and prolactin release: hypothalamic GABA and catecholamine turnover rates. Brain Res. 231: 353, 1982.
Melis G.B., Paoletti A.M., Mais V., Gambacciani M., Guarnieri G., Strigini F., Fruzzetti F., Fioretti P. Inhibitory effect of the dopamine agonist bromocriptine on the postcastration gonadotropin rise in women. J. Clin. Endocrinol. Metab. 53: 530, 1981.
Fioretti P., Guarnieri G., Mais V., Paoletti A.M., Gambacciani M., Fruzzetti F., Facchini V., Gargiulo T., Facchinetti F., Melis G.B. Short term effect of gonadectomy on pituitary secretion in females. In: Fioretti P., Martini L., Melis G.B., Yen S.S.C. (Eds.), The menopause: clinical, endocrinological and patho physiological aspects. Academic Press, London, 1982, p. 81.
Wang C.F., Lasley B.L., Lein A., Yen S.S.C. The functional changes of the pituitary gonadotrophs during the menstrual cycle. J. Clin. Endocrinol. Metab. 42: 718, 1976.
Banzan A.M., Donoso A.O. Failure of ovulation caused by GABA in the immature rat given pregnant mare serum gonadotropin. Endocrinology 110: 2180, 1982.
Elias A.N., Szekeres A.V., Stone S., Valenta L.J. A presumptive role for gamma-aminobutyric acid in the regulation of gonadotropin secretion in man. Am. J. Obstet. Gynecol. 144: 72, 1982.
Wang C.F., Lasley B.L., Yen S.S.C. Gonadotropin secretion in response to low and high doses of LRF in normal and hypogonadal women (Functional disparity of the gonadotrophs). J. Clin. Endocrinol. Metab. 42: 427, 1976.
Sar M., Stumpf W.E., Tappaz M.L. Localization of 3H-estradiol in preoptic gabaergic neurons. Fed. Proc. 42: 495, 1983 (Abstract).
Jennes L., Stumpf W.E., Tappaz M.L Anatomical relationships of dopaminergic and GABAergic systems with the GnRH-systems in the septohypothalamic area. Immunohistochemical studies. Exp. Brain Res. 50: 91, 1983.
Fuchs E., Mansky T., Stock K-W., Vijayan E., Wuttke W. Involvement of catecholamines and glutamate in GABAergic mechanism regulatory to luteinizing hormone and prolactin secretion. Neuroendocrinology 38: 434, 1984.
Elias A.N., Szekeres A.V., Stone S., Valenta L.J., Haw T., Ascher M.S. GABAergic and dopaminergic mechanisms in gonadotropin secretion in males: effects of baclofen and metoclopramide. Acta Endocrinol. (Kbh.) 103: 451, 1983.
Quigley M.E., Yen S.S.C. The role of endogenous opiates on LH secretion during the menstrual cycle. J. Clin. Endocrinol. Metab. 51: 179, 1980.
Ropert J.F., Quigley M.E., Yen S.S.C. Endogenous opiates modulate pulsatile luteinizing hormone release in humans. J. Clin. Endocrinol. Metab. 52: 583, 1981.
Rasmussen D.D., Liu J.H., Wolf P.L., Yen S.S.C. Endogenous opioid regulation of gonadotropin-releasing hormone release from the human fetal hypothalamus in vitro. J. Clin. Endocrinol. Metab. 57: 881, 1983.
Aronin N., Difiglia M., Graveland G.A., Schwartz W.J., Wu J-Y. Localization of immunoreactive enkephalin in GABA synthesizing neurons of the rat neostriatum. Brain Res. 300: 376, 1984.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Melis, G.B., Mais, V., Paoletti, A.M. et al. Involvement of endogenous gabaergic system in the modulation of gonadotropin secretion in normal cycling women. J Endocrinol Invest 9, 71–76 (1986). https://doi.org/10.1007/BF03348068
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03348068