Abstract
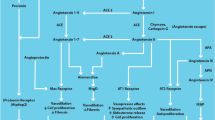
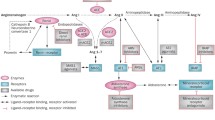
Recent studies suggest that a dysregulation of the aldosterone system is involved in the pathophysiology of different cardiovascular diseases, including myocardial failure and several cases of essential hypertension. In both rat models and in humans, aldosterone action has been shown to induce heart remodeling and interstitial and perivascular fibrosis of the myocardium. For these reasons, a rationale for the use of aldosterone antagonists (ARAs) of the spirolactone family, which have been available for decades in the treatment of aldosterone excess syndromes, has now emerged. Moreover, the recent validation of their use, in combination with the current therapy, for the treatment of these cardiovascular diseases by trials like the RALES Study has further strenghtened this approach. The development of compounds, like eplerenone, with a greater selectivity for mineralocorticoid receptors, seems promising also in terms of reduction of endocrine side effects. The addition of aldosterone antagonists to the conventional therapy of myocardial failure and of selected cases of hypertension thus appears beneficial, resulting in an improved survival rate and a reduced incidence of cardiac complications. This review article, after a brief recall of the physiology of the aldosterone system, addresses the emerging role of aldosterone in cardiovascular diseases, considers the pharmacology of ARAs and the novel therapeutical applications of these compounds in hypertension and heart failure.
Similar content being viewed by others
References
Jeunemaitre X, Kreft-Jais C, Chatellier G, et al. Long-term experience of spironolactone in essential hypertension. Kidney Int Suppl 1988, 26: S14–7.
Henry M, Wehrlen M, Pelletier B, Capron MH. Spironolactone versus nifedipine in essential hypertension. Am J Cardiol 1990, 65: 36K–8K.
Glorioso N, Tonolo G, Troffa C, et al. Recognition of markers of response to potassium-canrenoate in essential hypertension. Steroids 1995, 60: 105–9.
Campbell SE, Diaz-Arias AA, Weber KT. Fibrosis of the human heart and systemic organs in adrenal adenoma. Blood Press 1992, 1: 149–560.
Fullerton MJ, Funder JW. Aldosterone and cardiac fibrosis: in vitro studies. Cardiovasc Res 1994, 28: 1863–7.
Robert V, Van Thiem N, Cheav SL, et al. Increased cardiac Types 1 and 3 collagen mRNAs in aldosterone-salt hypertension. Hypertension 1994, 24: 30–6.
Young M, Fullerton M, Dilley R, Funder J. Mineralocorticoids, hypertension, and cardiac fibrosis. J Clin Invest 1994, 93: 2578–83.
Young MJ, Funder JW. The renin-angiotensin-aldosterone system in experimental mineralocorticoid-salt-induced cardiac fibrosis. Am J Physiol 1996, 271: E883–8.
Swedberg K, Eneroth P, Kjekshus J, Wilhelmsen L. Hormones regulating cardiovascular function in patients with severe congestive heart failure and their relation to mortality. CONSENSUS Trial Study Group. Circulation 1990, 82: 1730–6.
Duprez DA, Bauwens FR, De Buyzere ML, et al. Influence of arterial blood pressure and aldosterone on left ventricular hypertrophy in moderate essential hypertension. Am J Cardiol 1993, 71: 17A–20A.
Takeda R, Matsubara T, Miyamori I, Hatakeyama H, Morise T. Vascular complications in patients with aldosterone producing adenoma in Japan: comparative study with essential hypertension. The Research Committee of Disorders of Adrenal Hormones in Japan. J Endocrinol Invest 1995, 18: 370–3.
Shigematsu Y, Hamada M, Okayama H, et al. Left ventricular hypertrophy precedes other target-organ damage in primary aldosteronism. Hypertension 1997, 29: 723–7.
Saavedra JM. Brain and pituitary angiotensin. Endocr Rev 1992, 13: 329–80.
Murphy TJ, Alexander RW, Griendling KK, Runge MS, Bernstein KE. Isolation of a cDNA encoding the vascular Type-1 angiotensin II receptor. Nature 1991, 351: 233–6.
Reaux A, Fournie-Zaluski MC, Llorens-Cortes C. Angiotensin III: a central regulator of vasopressin release and blood pressure. Trends Endocrinol Metab 2001, 12: 157–62.
Mortensen RM, Williams GH. Aldosterone action, physiology. In: De Groot LJ ed. Endocrinology. Philadephia: W. B. Saunders, 1995, 1668–76.
Struthers AD. Evidence for myocardial synthesis of aldosterone producing myocardial fibrosis in man. Clin Sci (Lond) 2002, 102: 387.
Verrey F. Early aldosterone action: toward filling the gap between transcription and transport. Am J Physiol 1999, 277: F319–27.
Pearce D. The role of SGK1 in hormone-regulated sodium transport. Trends Endocrinol Metab 2001, 12: 341–7.
Verrey F, Kraehenbuhl JP, Rossier BC. Aldosterone induces a rapid increase in the rate of Na,K-ATPase gene transcriptin in cultured kidney cells. Mol Endocrinol 1989, 3: 1369–76.
Blot-Chabaud M, Wanstok F, Bonvalet J, Farman N. Cell sodium-induced recruitment of Na+-K+-ATPase pumps in rabbit cortical collecting tubules is aldosterone-dependent. J Biol Chem 1990, 265: 11676–81.
Ikeda U, Hyman R, Smith TW, Medford RM. Aldosteronemediated regulation of Na+-K+-ATPase gene expression in adult and neonatal rat cardiocytes. J Biol Chem 1991, 266: 12058–66.
Arriza JL, Weinberger C, Cerelli G, et al. Cloning of human mineralocorticoid receptor complementary DNA: structural and functional kinship with the glucocorticoid receptor. Science 1987, 237: 268–75.
Hollenberg SM, Weinberger C, Ong ES, et al. Primary structure and expression of a functional human glucocorticoid receptor cDNA. Nature 1985, 318: 635–41.
Kellendonk C, Gass P, Kretz O, Schutz G, Tronche F. Corticosteroid receptors in the brain: gene targeting studies. Brain Res Bull 2002, 57: 73–83.
Lombes M, Farman N, Bonvalet JP, Zennaro MC. Identification and role of aldosterone receptors in the cardiovascular system. Ann Endocrinol (Paris) 2000, 61: 41–6.
Funder JW, Marver D. Aldosterone action, biochemistry. In: De Groot LJ ed. Endocrinology. Philadelphia: W.B. Saunders, 1995, 1677–84.
Gomez-Sanchez EP, Fort CM, Gomez-Sanchez CE. Intracerebroventricular infusion of RU28318 blocks aldosterone- salt hypertension. Am J Physiol 1990, 258: E482–4.
Gomez-Sanchez EP, Venkataraman MT, Thwaites D, Fort C. ICV infusion of corticosterone antagonizes ICV-aldosterone hypertension. Am J Physiol 1990, 258: E649–53.
Christ M, Sippel K, Eisen C, Wehling M. Non-classical receptors for aldosterone in plasma membranes from pig kidneys. Mol Cell Endocrinol 1994, 99: R31–4.
Christ M, Douwes K, Eisen C., et al. Rapid effects of aldosterone on sodium transport in vascular smooth muscle cells. Hypertension 1995, 25: 117–23.
Meyer C, Christ M, Wehling M. Characterization and solubilization of novel aldosterone-binding proteins in porcine liver microsomes. Eur J Biochem 1995, 229: 736–40.
Wehling M. Specific, nongenomic actions of steroid hormones. Annu Rev Physiol 1997, 59: 365–93.
Connell JM, Fraser R, Davies E. Disorders of mineralocorticoid synthesis. Best Pract Res Clin Endocrinol Metab 2001, 15: 43–60.
Ferrari P, Bianchetti M, Frey FJ. Juvenile hypertension, the role of genetically altered steroid metabolism. Horm Res 2001, 55: 213–23.
Stowasser M, Gordon RD. Familial hyperaldosteronism. J Steroid Biochem Mol Biol 2001, 78: 215–29.
Ferrari P. Genetics of the mineralocorticoid system in primary hypertension. Curr Hypertens Rep 2002, 4: 18–24.
Edwards CRW. Primary mineralocorticoid excess. In: L. J. De Groot ed. Endocrinology. Philadelphia: W.B. Saunders, 1995, 1775–803.
Mantero F, Lucarelli G. Aldosterone antagonists in hypertension and heart failure. Ann Endocrinol (Paris) 2000, 61: 52–60.
Lim PO, Macdonald TM, Holloway C, et al. Variation at the aldosterone synthase (CYP11B2) locus contributes to hypertension in subjects with raised aldosterone-to-renin ratio. J Clin Endocrinol Metab 2002, 87: 4398–402.
Williams GH, Moore TJ. Hormonal aspects of hypertension. In: De Groot LJ ed. Endocrinology. Philadelphia: W.B. Saunders, 1995, 2917–34.
Stier CT Jr, Chander PN, Rocha R. Aldosterone as a mediator in cardiovascular injury. Cardiol Rev 2002, 10: 97–107.
Mizuno Y, Yoshimura M, Yasue H, et al. Aldosterone production is activated in failing ventricle in humans. Circulation 2001, 103: 72–7.
Satoh M, Nakamura M, Saitoh H, et al. Aldosterone synthase (CYP11B2) expression and myocardial fibrosis in the failing human heart. Clin Sci (Lond) 2002, 102: 381–6.
Weber KT, Brilla CG. Pathological hypertrophy and cardiac interstitium. Fibrosis and renin-angiotensin-aldosterone system. Circulation 1991, 83: 1849–65.
Brilla CG, Pick R, Tan LB, Janicki JS, Weber KT. Remodeling of the rat right and left ventricles in experimental hypertension. Circ Res 1990, 67: 1355–64.
Brilla CG, Matsubara LS, Weber KT. Antifibrotic effects of spironolactone in preventing myocardial fibrosis in systemic arterial hypertension. Am J Cardiol 1993, 71: 12A–6A.
Abraham WT, Schrier RW. Edematous disorders: pathophysiology of renal sodium and water retention and treatment with diuretics. Curr Opin Nephrol Hypertens 1993, 2: 798–805.
Kagawa CM, Cella JA, Van Arman CG. Action of new steroids in blocking effects of aldosterone and deoxycorticosterone on salt. Science 1957, 126: 1015–6.
Fanestil DD Mechanism of action of aldosterone blockers. Semin Nephrol 1988, 8: 249–63.
Overdiek HW, Merkus FW. The metabolism and biopharmaceutics of spironolactone in man. Rev Drug Metab Drug Interact 1987, 5: 273–302.
Armanini D, Karbowiak I, Goi A, Mantero F, Funder JW. In-vivo metabolites of spironolactone and potassium canrenoate: determination of potential anti-androgenic activity by a mouse kidney cytosol receptor assay. Clin Endocrinol (Oxf) 1985, 23: 341–7.
Jackson EK. Diuretics. In: Hardman JG, Limbird LE and Gilman AG eds. Goodman & Gilman’s the Pharmacological Basis of Therapeutics. New York: McGraw-Hill, 2001, 757–87.
Dupont A. Disappearance of spironolactone-induced gynaecomastia during treatment with potassium canrenoate. Lancet 1985, 2: 731.
McMahon EG. Recent studies with eplerenone, a novel selective aldosterone receptor antagonist. Curr Opin Pharmacol 2001, 1: 190–6.
de Gasparo M, Joss U, Ramjoue HP, et al. Three new epoxy-spirolactone derivatives: characterization in vivo and in vitro. J Pharmacol Exp Ther 1987, 240: 650–6.
de Gasparo M, Whitebread SE, Preiswerk G, et al. Antialdosterones: incidence and prevention of sexual side effects. J Steroid Biochem 1989, 32: 223–7.
Mantero F, Opocher G, Rocco S, Carpene G, Armanini D. Long-term treatment of mineralocorticoid excess syndromes. Steroids 1995, 60: 81–6.
Staessen J, Lijnen P, Fagard R, Verschueren LJ, Amery A. Rise in plasma concentration of aldosterone during long-term angiotensin II suppression. J Endocrinol 1981, 91: 457–65.
McKelvie RS, Yusuf S, Pericak D, et al. Comparison of candesartan, enalapril, and their combination in congestive heart failure: randomized evaluation of strategies for left ventricular dysfunction (RESOLVD) pilot study. The RESOLVD Pilot Study Investigators. Circulation 1999, 100: 1056–64.
Weinberger MH, Roniker B, Krause SL, Weiss RJ. Eplerenone, a selective aldosterone blocker, in mild-to-moderate hypertension. Am J Hypertens 2002, 15: 709–16.
Anand IS, Ferrari R, Kalra GS, et al. Edema of cardiac origin. Studies of body water and sodium, renal function, hemodynamic indexes, and plasma hormones in untreated congestive cardiac failure. Circulation 1989, 80: 299–305.
Bayliss J, Norell M, Canepa-Anson R, Sutton G, Poole-Wilson P. Untreated heart failure: clinical and neuroendocrine effects of introducing diuretics. Br Heart J 1987, 57: 17–22.
The Consensus Trial study Group. Effect of enalapril on mortality in severe congestive heart failure: results of the Cooperative North Scandinavian Enalapril Survival Study (Consensus). N Engl J Med 1987, 316: 1429–35.
The Solvd Investigators. Effect of enalapril on survival in patients with reduced left ventricular ejection fraction and congestive heart failure. N Engl J Med 1991, 325: 293–302.
Swedberg K, Eneroth P, Kjekshus J, Wilhelmsen L. Hormones regulating cardiovascular function in patients with severe congestive heart failure and their relation to mortality. CONSENSUS Trial Study Group. Circulation 1990, 82: 1730–6.
Vantrimpont P, Rouleau JL, Ciampi A, et al. Two-year time course and significance of neurohumoral activation in the Survival and Ventricular Enlargement (SAVE) Study. Eur Heart J 1998, 19: 1552–63.
Struthers AD. Aldosterone escape during angiotensin-converting enzyme inhibitor therapy in chronic heart failure. J Card Fail 1996, 2: 47–54.
Zannad F. Angiotensin-converting enzyme inhibitor and spironolactone combination therapy. New objectives in congestive heart failure treatment. Am J Cardiol 1993, 71: 34A–9A.
Pitt B, Zannad F, Remme WJ, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators. N Engl J Med 1999, 341: 709–17.
Rocha R, Stier CT Jr, Kifor I, et al. Aldosterone: a mediator of myocardial necrosis and renal arteriopathy. Endocrinology 2000, 141: 3871–8.
Martinez DV, Rocha R, Matsumura M, et al. Cardiac damage prevention by eplerenone: comparison with low sodium diet or potassium loading. Hypertension 2002, 39: 614–8.
Pitt B, Williams G, Remme W, et al. The EPHESUS trial: eplerenone in patients with heart failure due to systolic dysfunction complicating acute myocardial infarction. Eplerenone Post-AMI Heart Failure Efficacy and Survival Study. Cardiovasc Drugs Ther 2001, 15: 79–87.
Villa GL, Barletta G, Romanelli RG, et al. Cardiovascular effects of canrenone in patients with preascitic cirrhosis. Hepatology 2002, 35: 1441–8.
Farquhar C, Lee O, Toomath R, Jepson R. Spironolactone versus placebo or in combination with steroids for hirsutism and/or acne. Cochrane Database Syst Rev 2001, 4: CD000194.
Helfer EL, Miller JL, Rose LI. Side-effects of spironolactone therapy in the hirsute woman. J Clin Endocrinol Metab 1988, 66: 208–11.
Funder JW. Aldosterone resurgens-letter from EPHESUS. J Clin Endocrinol Metab 2003, 88: 2373–5.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Magni, P., Motta, M. Aldosterone receptor antagonists: Biology and novel therapeutical applications. J Endocrinol Invest 26, 788–798 (2003). https://doi.org/10.1007/BF03347366
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03347366