Abstract
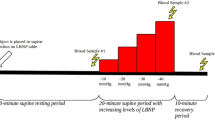
Typical modifications of cardiovascular activity and water and salt homeostasis throughout female reproductive life are well known. Differences in plasma levels of calcitonin gene-related peptide (CGRP) and atrial natriuretic peptide (ANP) have been observed in conditions characterized by different estrogenic levels, suggesting a correlation between female reproductive function and these cardiovascular hormones. The aim of our study was to investigate in hypothalamic amenorrhea the relationship between estrogen deficiency and plasma ANP and CGRP response to adaptive tests (saline infusion test and upright posture test, respectively). Women with hypothalamic amenorrhea (aged 18–28 years) (n=6) and age-matched healthy controls (n=6) underwent both functional tests. Plasma CGRP and ANP levels were measured by specific radioimmunoassays before and in course of the tests. Basal plasma CGRP levels of amenorrheic patients did not significantly differ from those of normal women, while basal plasma ANP levels were significantly higher compared to controls (p<0.01). In amenorrheic women, plasma CGRP levels showed a significant increase in response to upright posture test, though lower than the increase observed in normal women. In contrast, saline infusion test determined a significant increase in plasma ANP levels only in control subjects. In women with hypothalamic amenorrhea, the altered response of CGRP and ANP to adaptive stimuli indicates a partial derangement in the control of the secretion of these cardiovascular hormones. Nevertheless, the differences between such modifications and those observed in other conditions of altered estrogenic levels, suggest that in amenorrheic women hypogonadism is not the major factor influencing CGRP and ANP response to adaptive stimuli.
Similar content being viewed by others
References
Longo L. Maternal blood volume and cardiac output during pregnancy: a hypothesis of endocrinological control. Am. J. Physiol. 245: R720, 1983.
Valenzuela G., Brace R., Longo L. Lymphatic and vascular response to fluid infusion in castrated and non-castatred sheep. Am J. Physiol. 252: R1114, 1997.
De Bold A., Borestein H., Veress A.T., Sonnenberg H. A rapid and potent natriuretic response to intravenous injection of atrial myocardial extracts in rats. Life Sci 28: 89, 1981.
Oshima T., Currie M., Geller D., Needleman P. An atrial peptide is a potent renal vasodilator substance. Circ. Res. 154: 612, 1984.
Nicholls M., Richards A. Human studies with atrial natriuretic factor. Endocrin. Metab. Clin. North Am. 16: 19, 1987.
Ruskoaho H. Atrial natriuretic peptide: synthesis, release and metabolism. Pharmacol. Rev. 44: 479, 1992.
Espiner E. Physiology of natriuretic peptide. J Int. Med. 235: 527, 1994.
Kimura T., Abe K., Ota K., Omata K., Shoji M., Kudo K., Matsui K., Inoue M., Yasujima M., Yoshinaga K. Effects of acute water load, hypertonic saline infusion, and furosemide administration on atrial natriuretic peptide and vasopressin release in humans. J. Clin. Endocrinol. Metab. 62: 1003, 1986.
Clark B., Elahi D., Epstein F. The influence of gender, age and menstrual cycle on plasma atrial natriuretic peptide. J. Clin. Endocrinol. Metab. 70: 349, 1990.
Otzuki Y., Okamoto E., Iwata I., Nishino E., Mitsuda N., Mori M., Tagaki T., Sugita N., Tanizawa O. Changes in concentration of human atrial natriuretic peptide in normal pregnancy and toxaemia. J. Endocrinol. 114: 325, 1987.
Merkouris R.W., Miller F.C., Catanzarite V., Rigg L.A., Quirk J.G., Vesely D.L. Increase in the plasma levels of the N-terminal and C-terminal portions of the prohormone of atrial natriuretic factor during normal pregnancy. Am J. Obstet. Gynecol. 162: 859, 1990.
Yoshimura T., Yoshimura M., Yasue H. Plasma concentration of atrial natriuretic peptide during normal pregnancy and the postpartum period. J. Endocrinol. 140: 393, 1994.
Tshopp F., Henke H., Peterman J. Calcitonin gene-related peptide and its binding sites in the human central nervous system and pituitary. Proc. Natl. Acad. Sci. USA 82: 248, 1997.
Mulderry P., Ghatei M., Rodrigo K., Allen J.M., Rosenfeld M.G., Polak J.M., Bloom S.A. Calcitonin gene-related peptide in cardiovascular tissue of the rat. Neuroscience 14: 947, 1985.
Trasforini G., Margutti A., Vergnani L., Ambrosio M., Valentini A., Rossi R., Portaluppi F. Evidence that enhancement of cholinergic tone increases basal plasma levels of calcitonin gene-related peptide in normal man. J. Clin. Endocrinol. Metab. 78: 763, 1994.
Trasforini G., Margutti A., Valentini A., Rossi R., Degli Uberti E.C. Interrelationship between calcitonin gene-related peptide and sympathoadrenomedullary system: effects of administration of epinephrine and norepinephrine in healthy men. Regul. Pept. 63: 56, 1996.
Portaluppi F., Vergnani, I., Margutti A., Ambrosio M.R., Bondanelli M., Trasforini G., Rossi R., Degli Liberti E.C. Modulatory effect of the renin-angiotensin system on the plasma levels of calcitonin gene-related peptide in normal man. J. Clin. Endocrinol. Metab., 177: 816, 1993.
Girgis S., MacDonald D., Stevenson J., Bevis P.J., Lynch C., Wimalawansa S.J., Self C.H., Morris H.R., MacIntyre I. Calcitonin gene-related peptide: a potent vasodilator and major product of calcitonin gene. Lancet 2: 14, 1985.
Vesely M., Overton R.M., McCormick M.T., Schocken D.D. Atrial natriuretic peptide increase calcitonin gene-related peptide within human circulation. Metabolism 46: 818, 1997.
Valentini A., Petraglia F., De Vita D., Nappi C., Margutti A., Degli Liberti E. C., Genazzani A.R. Changes in plasma calcitonin gene-related peptide in postmenopausal women. Am. Obstet. Gynecol. 175: 638, 1996.
Baranowska B., Wasilewska-Dziubinska E., Raditzikowska M., Plonowski A., Roguski K. Impaired response of atrial natriuretic peptide to acute water load in obesity and in anorexia nervosa. Eur. J. Endocrinol. 132: 147, 1995.
Brewerton T. Toward a unified theory of serotonin dysregulation in eating and related disorders. Psychoneuroendocrinology 20: 561, 1995.
Leibowitz S. Neurochemical-neuroendocrine systems in the brain controlling macronutrient intake and metabolism. Trends Neurosci. 15: 491, 1992.
Genazzani A.D., Gastaldi M., Volpe M., Petraglia F., Genazzani A.R. Spontaneous episodic release of adenohypophyseal hormones in hypothalamic amenorrhea. Gynecol. Endocrinol. 9: 325, 1995.
Yen S.S. Female hypogonadotropic hypogonadism. Hypothalamic amenorrhea syndrome. Endocrinol. Metab. Clin. North Am. 22: 29, 1997.
Golden N., Shenker J. Amenorrhea in anorexia nervosa: neuroendocrine control of hypothalamic disfunction. Intern. J. Eating. Dis. 16: 53, 1994.
Nappi R., Petraglia F., Genazzani A., D’Ambrogio G., Zara C., Genazzani A.R. Hypothalamic amenorrhea: evidence for a central derangement of hypothalamic-pituitary-adrenal cortex axis activity. Fertil. Steril. 59: 57, 1993.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bernardi, F., Valentini, A., Margutti, A. et al. Hypothalamic amenorrhea and cardiovascular hormones: Changes of plasma calcitonin gene-related peptide and atrial natriuretic peptide levels. J Endocrinol Invest 21, 251–256 (1998). https://doi.org/10.1007/BF03347311
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03347311