Abstract
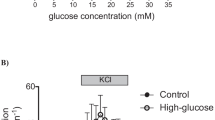
We investigated the effect of 24 h exposure to 100 nmol/l glibenclamide on insulin secretion in isolated rat pancreatic islets. The insulin content was similar in control islets and in islets preincubated with 100 nmol/l glibenclamide for 24 h. In islets preexposed to glibenclamide: 1) the subsequent response to a maximal glibenclamide stimulatory concentration (10 µmol/l, 1 h at 37 C) was greatly reduced in comparison to control islets (0.69 ± 0.20 % vs 2.16 ± 0.41%; mean ± SE; n=14; p < 0.001); 2) the response to 100 µmol/l tolbutamide stimulation was also reduced (0.55 ± 0.15% vs 2.38 ± 0.44 %; n=8; p < 0.001); 3) the response to 16.7 mmo/l glucose, both in the presence or in the absence of 1 mmol/l IBMX, a phosphodiesterase inhibitor, was also diminished by about 50% (1.79 ± 0.39% vs. 3.22 ± 0.42%; n= 14, p < 0.001). In glibenclamide pretreated islets, blunted responses to stimuli were confirmed also by dynamic studies using a perifusion system. The effect of glibenclamide preincubation was fully reversible: when islets cultured in the presence of glibenclamide were transferred to a glibenclamide-free medium for further 24 h, insulin release in response to glibenclamide stimulation returned to control values. We conclude that prolonged exposure of rat pancreatic islets to glibenclamide induces a reversible desensitization to a variety of metabolic stimuli. The inhibition by prolonged glibenclamide exposure of a common pathway in the mechanism of insulin release is one possible explanation for these results.
Similar content being viewed by others
References
DeFronzo R.A., Simonson D.C. Oral sulfonylurea agents suppress hepatic glucose production in non-insulin-dependent diabetic individuals. Diabetes Care 7(Suppl 1): 72, 1984.
Pfeiffer M.A., Halter J.B., Beard J.C., Judewitsch R., Porte D. Insulin responses to nonglucose stimuli in non-insulin-dependent diabetes mellitus during a tolbutamide infusion. Diabetes 31: 154, 1982.
Duckworth W.C., Solomon S.S., Kitabchi A.E. Effect of chronic sulfonylurea therapy on plasma insulin and proinsulin levels. J. Clin. Endocrinol. Metab. 35: 585, 1972.
Fineberg S.E., Schneider S.H. Glipizide versus tolbutamide, an open trial: the effect on insulin secretory patterns and glucose concentrations. Diabetologia 18: 49, 1980.
Karam J.H., Sanz E., Salomon E., Nolte M.S. Selective unresponsiveness of pancreas β-cells to acute sulfonylurea stimulation during sulfonylurea therapy in NIDDM. Diabetes 35: 1314, 1986.
Wajchenberg B.L., Nery M., Leme C.E., Silveira A.A., Fioratti P., Germek O.A. Effect of prolonged gliclazide treatment on blood and plasma insulin response in obese patients with maturity-onset diabetes. Clin.Pharmacol.Ther., 27: 375, 1980.
Hosker J.P., Burnett M.A., Davies E.G., Harris E.A., Turner R.C. Sulphonylurea therapy doubles B-cell response to glucose in type 2 diabetic patients. Diabetologia 28: 809, 1985.
Filipponi P., Marcelli M., Nicoletti I., Pacifici R., Santeusanio F., Brunetti P. Suppressive effect of long-term sulfonylurea treatment on A, B, and D cells of normal rat pancreas. Endocrinology 113: 1972, 1983.
Dunbar J.C., Foà P.P. An inhibitory effect of tolbutamide and glibenclamide on the pancreatic islets of normal animals. Diabetologia 10: 27, 1974
Schauder P., Arends J., Freichs H. Onset and reversibility of changes in secretory function and composition of isolated rat pancreatic islets following long-term administration of high or low tolbutamide doses. Metabolism 26: 9, 1977.
Purrello F., Vetri M., Gatta C., Gullo D., Vigneri R. Effects of high glucose on insulin secretion by isolated rat islets and purified β-cells and possible role of glycosylation. Diabetes 38: 1417, 1989.
Groop L., Groop P.H., Stenman S., Saloranta C., Totterman K.J., Fyhrquist F, Melander A. Comparison of pharmacokinetics, metabolic effects and mechanisms of action of glyburide and glipizide during long-term treatment. Diabetes Care 10: 671, 1987.
Pipeleers D.G., In’t Veld P.A., Maes E., Van De Winkel M. Glucose-induced insulin release depends on functional cooperation between islet cells. Proc. Natl. Acad. Sci USA 79: 7322, 1982.
Bolaffi J.L., Bruno L., Heidt A., Grodsky G.M. Characteristics of desensitization of insulin secretion in fully in vitro systems. Endocrinology 122: 1801, 1988.
Hoenig L.C., MacGregor L.C., Matschinsky P.M. In vitro exhaustion of pancreatic β-cells. Am. J. Physiol. 250: E502, 1986.
Grodsky G.M. A new phase of insulin secretion: how will it contribute to our understanding of β-cell function? Diabetes 38: 673, 1989.
Henquin J.C. Tolbutamide stimulation and inhibition of insulin release: studies of the underlying ionic mechanisms in isolated rat islets. Diabetologia 18: 151, 1980.
Zawalich W.S. Phosphoinoside hydrolysis and insulin secretion in response to glucose are impaired in isolated rat islets by prolonged exposure to the sulfonylurea tolbutamide. Endocrinology 125: 281, 1989.
Robertson R.P., Porte D. The glucose-receptor: a defective mechanism in diabetes mellitus distinct from the beta adrenergic receptor. J. Clin. Invest. 2: 870, 1976
Palmer J.P., Benson J.W., Walter R.M., Ensinck J.W. Arginine-stimulated acute phase of insulin and glucagon secretion in diabetic subjects. J. Clin. Invest. 8: 565, 1976.
Gorus F.K, Schuit F.C., In’t Veld P.A., Gepts W., Pipeleers D.G. Interaction of sulfonylureas with pancreatic β-cells. A study with glyburide. Diabetes 37: 1090, 1988.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gullo, D., Rabuazzo, A.M., Vetri, M. et al. Chronic exposure to glibenclamide impairs insulin secretion in isolated rat pancreatic islets. J Endocrinol Invest 14, 287–291 (1991). https://doi.org/10.1007/BF03346813
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03346813