Abstract
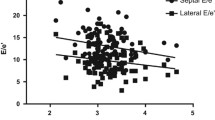
Objective: To evaluate the morpho-functional changes of the myocardium in patients with primary aldosteronism (PA). Design: An observational study in a university referral center for blood pressure diseases. Patients: Twenty-three patients with PA, 24 patients with essential hypertension (EH), and 15 normotensive controls (C) underwent conventional echocardiography with integrated backscatter (IBS) and tissue Doppler imaging (TDI) analysis. The corrected IBS (C-IBS) values and the systo-diastolic variation of IBS (CV-IBS) were performed at both interventricular septum and the posterior wall levels. TDI myocardial systolic (Sm), early diastolic (Em), and late diastolic (Am) velocities of both left ventricular walls were also determined. Results: In PA patients, septal and posterior wall CV-IBS were significantly lower than C (p<0.0001) and EH patients (p<0.001). In EH, CV-IBS was significantly lower than C (p<0.001). Patients with PA exhibited lower Sm, lower Em, and higher Am, and a subsequently reduced Em/Am ratio than C (p<0.001 for all) and EH (p<0.01 for all) at interventricular septum and lateral wall levels. In the latter, Sm, Em, and Em/Am ratio were lower and Am was higher than C (p<0.001 for all). In PA and EH patients, CV-IBS at both septum (r=−0.66, p<0.001) and posterior wall levels (r=−0.67, p<0.001) and Sm peak of both septum (r=−0.52, p<0.001) and lateral wall (r=−0.55, p<0.001) were inversely related to plasma aldosterone. Conclusions: Patients with PA showed myocardial wall remodeling characterized by increased myocardial fibrosis and early left ventricular systodiastolic function abnormalities.
Similar content being viewed by others
References
Mano A, Tatsumi T, Shiraishi J, et al. Aldosterone directly induces myocyte apoptosis through calcineurin-dependent pathways. Circulation 2004, 110: 317–23.
Lijnen P, Petrov V. Induction of cardiac fibrosis by aldosterone. J Mol Cell Cardiol 2000, 32: 865–79.
Burniston JG, Saini A, Tan LB, Goldspink DF. Aldosterone induces myocyte apoptosis in the heart and skeletal muscles of rats in vivo. J Mol Cell Cardiol 2005, 39: 395–9.
Schmidt BM, Schmieder RE. Aldosterone-induced cardiac damage: focus on blood pressure independent effects. Am J Hypertens 2003, 16: 80–6.
Rickard AJ, Funder JW, Fuller PJ, Young MJ. The role of the glucocorticoid receptor in mineralocorticoid/salt-mediated cardiac fibrosis. Endocrinology 2006, 147: 5901–6.
Susic D, Varagic J, Ahn J, Matavelli L, Frohlich ED. Long-term mineralocorticoid receptor blockade reduces fibrosis and improves cardiac performance and coronary hemodynamics in elderly SHR. Am J Physiol Heart Circ Physiol 2007, 292: H175–9.
Goodfriend TL. Aldosterone: a hormone of cardiovascular adaptation and maladaptation. J Clin Hypertens (Greenwich) 2006, 8: 133–9.
Ward MR, Kanellakis P, Ramsey D, Funder J, Bobik A. Eplerenone suppresses constrictive remodeling and collagen accumulation after angioplasty in porcine coronariy arteries. Circulation 2001, 104: 467–72.
Rocha R, Stier CT Jr. Pathophysiological effects of aldosterone in cardiovascular tissues. Trends Endocrinol Metab 2001, 12: 308–14.
Rossi G, Boscaro M, Ronconi V, Funder JW. Aldosterone as a cardiovascular risk factor. Trends Endocrinol Metab 2005, 16: 104–7.
Catena C, Colussi G, Lapenna R, et al. Long-term cardiac effects of adrenalectomy or mineralcorticoid antagonists in patients with primary aldosteronism. Hypertension 2007, 50: 911–8.
Stowasser M. New perspectives on the role of aldosterone excess in cardiovascular disease. Clin Exp Pharmacol Physiol 2001, 28: 783–91.
Milliez P, Girerd X, Plouin PF, Blacher J, Safar ME, Mourad JJ. Evidence for an increased rate of cardiovascular events in patients with primary aldosteronism. J Am Coll Cardiol 2005, 45: 1243–8.
Stowasser M, Sharman J, Leano R, et al. Evidence for abnormal left ventricular structure and function in normotensive individuals with familial hyperaldosteronism type I. J Clin Endocrinol Metab 2005, 90: 5070–6.
Catena C, Colussi G, Nadalini E, et al. Cardiovascular outcomes in patients with primary aldosteronism after treatment. Arch Intern Med 2008, 168: 80–5.
Hoyt RM, Skorton DJ, Collins SM, Melton HE Jr. Ultrasonic backscatter and collagen in normal ventricular myocardium. Circulation 1984, 69: 775–82.
Yalcin F, Kaftan A, Muderrisoglu H, et al. Is Doppler tissue velocity during early left ventricular filling preload independent? Heart 2002, 87: 336–9.
Palka P, Lange A, Fleming AD, Sutherland GR, Fenn LN, McDicken WN. Doppler tissue imaging: myocardial wall motion velocities in normal subjects. J Am Soc Echocardiogr 1995, 8: 659–68.
Oki T, Tabata T, Yamada H, et al. Clinical application of pulsed Doppler tissue imaging for assessing abnormal left ventricular relaxation. Am J Cardiol 1997, 79: 921–8.
Bernini G, Moretti A, Orlandini C, et al. Plasma and urine aldosterone to plasma renin activity ratio in the diagnosis of primary aldosteronism. J Hypertens 2008, 26: 981–8.
Mancia G, De Backer G, Dominiczak A, et al; The task force for the management of arterial hypertension of the European Society of Hypertension, The task force for the management of arterial hypertension of the European Society of Cardiology. 2007 Guidelines for the management of arterial hypertension: The Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). Eur Heart J 2007, 28: 1462–536.
Henry WL, DeMaria A, Gramiak R, et al. Report of the American Society of Echocardiography Committee on nomenclature and standards in two-dimensional echocardiography. Circulation 1980, 62: 212–7.
Devereux RB, Reichek N. Echocardiographic determination of left ventricular mass in man. Anatomic validation of the method. Circulation 1977, 55: 613–8.
Devereux RB, Lutas EM, Casale PN, et al. Standardization of M-mode echocardiographic left ventricular anatomic measurements. J Am Coll Cardiol 1984, 4: 1222–30.
Lang RM, Bierig M, Devereux RB, et al; American Society of Echocardiography’s Nomenclature and Standards Committee; Task Force on Chamber Quantification; American College of Cardiology Echocardiography Committee; American Heart Association; European Association of Echocardiography, European Society of Cardiology. Recommendations for chamber quantification. Eur J Echocardiogr 2006, 7: 79–108.
Franzoni F, Galetta F, Cupisti A, Rolla M, Santoro G, Pentimone F. Ultrasonic tissue characterization of the myocardium in anorexia nervosa. Acta Paediatr 2003, 92: 297–300.
Díez J. Effects of aldosterone on the heart: beyond systemic hemodynamics? Hypertension 2008, 52: 462–4.
Denolle T, Chatellier G, Julien J, Battaglia C, Luo P, Plouin PF. Left ventricular mass and geometry before and after etiologic treatment in renovascular hypertension, aldosterone-producing adenoma, and pheochromocytoma. Am J Hypertens 1993, 6: 907–13.
Rossi GP, Sacchetto A, Pavan E, et al. Remodeling of the left ventricle in primary aldosteronism due to Conn’s adenoma. Circulation 1997, 95: 1471–8.
Muiesan ML, Salvetti M, Paini A, et al. Inappropriate left ventricular mass in patients with primary aldosteronism. Hypertension 2008 Sep, 52: 529–34.
Hall CS, Scott MJ, Lanza GM, Miller JG, Wickline SA. The extracellular matrix is an important source of ultrasound backscatter from myocardium. J Acoust Soc Am 2000, 107: 612–9.
Mimbs JW, O’Donnell M, Bauwens D, Miller JW, Sobel BE. The dependence of ultrasonic attenuation and backscatter on collagen content in dog and rabbit hearts. Circ Res 1980, 47: 49–58.
Picano E, Pelosi G, Marzilli M, et al. In vivo quantitative ultrasonic evaluation of myocardial fibrosis in humans. Circulation 1990, 81: 58–64.
Wickline SA, Thomas LJ 3rd, Miller JG, Sobel BE, Perez JE. A relationship between ultrasonic integrated backscatter and myocardial contractile function. J Clin Invest 1985, 76: 2151–60.
Nozaki S, DeMaria AN, Helmer GA, Hammond HK. Detection of regional left ventricular dysfunction in early pacing-induced heart failure using ultrasonic integrated backscatter. Circulation 1995, 92: 2675–82.
Kozàkovà M, Buralli S, Palombo C, et al. Myocardial ultrasonic backscatter in hypertension: relation to aldosterone and endothe-lin. Hypertension 2003, 41: 230–6.
Rossi GP, Di Bello V, Ganzaroli C, et al. Excess aldosterone is associated with alterations of myocardial texture in primary aldosteronism. Hypertension 2002, 40: 23–7.
Kitamura M, Shimizu M, Ino H, et al. Collagen remodeling and cardiac dysfunction in patients with hypertrophic cardiomyopathy: the significance of type III and VI collagens. Clin Cardiol 2001, 24: 325–9.
Swedberg K, Eneroth P, Kjekshus J, Wilhelmsen L. Hormones regulating cardiovascular function in patients with severe congestive heart failure and their relation to mortality. CONSENSUS Trial Study Group. Circulation 1990, 82: 1730–6.
Tsioufis C, Tsiachris D, Dimitriadis K, et al. Myocardial and aortic stiffening in the early course of primari aldosteronism. Clin Cardiol 2008, 31: 431–6.
Gulati VK, Katz WE, Follansbee WP, Gorcsan J 3rd. Mitral annular descent velocity by tissue Doppler echocardiography as an index of global left ventricular function. Am J Cardiol 1996, 77: 979–84.
Yip GW, Zhang Y, Tan PY, et al. Left ventricular long-axis changes in early diastole and systole: impact of systolic function on diastole. Clin Sci (Lond) 2002, 102: 515–22.
Shimizu G, Hirota Y, Kita Y, Kawamura K, Saito T, Gaasch WH. Left ventricular midwall mechanics in systemic arterial hypertension. Myocardial function is depressed in pressure-overload hypertrophy. Circulation 1991, 83: 1676–84.
Palmes PP, Masuyama T, Yamamoto K, et al. Myocardial longitudinal motion by tissue velocity imaging in the evaluation of patients with myocardial infarction. J Am Soc Echocardiogr 2000, 13: 818–26.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Galetta, F., Bernini, G., Franzoni, F. et al. Cardiac remodeling in patients with primary aldosteronism. J Endocrinol Invest 32, 739–745 (2009). https://doi.org/10.1007/BF03346529
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03346529