Abstract
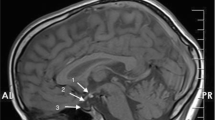
The authors present the case history of a 52-yr-old male patient with a unique association of combined pituitary hormone deficiency (CPHD) and situs inversus totalis. Except for signs and symptoms of pituitary hormone deficiency, the patient had no dysmorphic features, and hearing impairment, primary mental or neurologicaldefects were also absent. Pituitary magnetic resonance imaging (MRI) scan showed hypoplasia of the anterior lobe of the pituitary gland and an ectopic posterior pituitary lobe. Despite the presence of situs inversus totalis, thepatient was right-handed and functional MRI demonstrated left-hemisphere activation during language tests. Kartagener syndrome was considered, but immunofluorescence analysis showed normal localization of the outer dynein arm protein in respiratory epithelial cells obtained from the nasal mucosa. Direct DNA sequencing of all coding exons of the pituitary transcription factor 1 (PIT1) and prophet of PIT1 (PROP1) genes failed to detect disease-causing mutations, suggesting that these genes were not involved in the development of CPHD in our patient. More interestingly, the potential role of the paired like homeodomain transcription factor 2 (PITX2) gene, which has been implicated not only in CPHD, but also in left-right patterning in animal models, was also excluded, as sequencing showed the absence of mutations in coding exons of this gene. To our knowledge, PITX2 gene mutations have not been investigated in CPHD patients who had situs inversus totalis. We conclude that in contrast to animal models, the PITX2 gene is not involved in the development of situs inversus totalis, at least not in our CPHD patient.
Similar content being viewed by others
References
Kartagener M, Horlacher A. Situs viscerum inversus und polyposis nasi in einem Falle Familiaerer Bronchiektasien. Beitr Klin Tuberk 1936, 87: 331–3.
Afzelius BA. A human syndrome caused by immobile cilia. Science 1976, 193: 317–9.
Kurtoglu S, Atabek ME. Growth hormone deficiency, situs inversus, hypertrichosis and brachydactyly. J Pediatr Endocrinol Metab 2003, 16: 795–8.
Chabrolle JP, Labenne M, Cailliez D, Poinsot J, Bruel H, Vercoustre L. Hypoglossia, situs inversus and absence of the pituitary in the neonate: teratogenic effect of maternal hyperthermia? Arch Pediatr 1998, 5: 163–6.
Giacchino M, Toso P, Buzzi M, Nigro N, Benso L. Su di una rarissima associazione di processi morbosi. Minerva Pediatr 1977, 29: 1879–88.
Fliegauf M, Olbrich H, Horváth J, et al. Mislocalization of DNAH5 and DNAH9 in respiratory cells from patients with primary ciliary dyskinesia. Am J Respir Crit Care Med 2005, 171: 1343–9.
Deladoey J, Flück C, Büyükgebiz A, et al. “Hot spot” in the PROP1 gene responsible for combined pituitary hormone deficiency. J Clin Endocrinol Metab 1999, 84: 1645–50.
Malvagia S, Poggi GM, Pasquini E, et al. The de novo Q167K mutation in the POU1F1 gene leads to combined pituitary hormone deficiency in an Italian patient. Pediatr Res 2003, 54: 635–40.
Skaletsky HJ. Primer3 on the WWW for general users and for biologist programmers. In: Krawetz S, Misener S eds. Bioinformatics and methods and protocols: methods in molecular biology. Humana Press, Totowa NJ, 2000, 365–86.
Cohen LE. Genetic regulation of the embryology of the pituitary gland and somatotrophs. Endocrine 2000, 12: 99–106.
Cohen LE, Radovick S. Molecular basis of combined pituitary hormone deficiencies. Endocr Rev 2002, 23: 431–42.
Semina EV, Reiter R, Leysens NJ, et al. Cloning and characterization of a novel bicoid-related homeobox transcription factor gene, RIEG, involved in Rieger syndrome. Nat Genet 1996, 14: 392–9.
Muncke N, Niesler B, Roeth R, et al. Mutational analysis of the PITX2 coding region revealed no common cause for transposition of the great arteries (dTGA). BMC Med Genet 2005, 6: 20.
Franco D, Campione M, Kelly R, et al. Multiple transcriptional domains, with distinct left and right components, in the atrial chambers of the developing heart. Circ Res 2000, 87: 984–91.
Capdevila I, Belmonte JC. Knowing left from right: the molecular bases in lateral defects. Mol Med Today 2000, 6: 112–8.
Levin M. Left-right asymmetry in embryonic development: a comprehensive review. Mech Dev 2005, 122: 3–25.
Barth KA, Miklosi A, Watkins J, Bianco IH, Wilson SW, Adrew RJ. FSI zebrafish show concordant reversal of laterality of viscera, neuroanatomy, and a subset of behavioral responses. Curr Biol 2005, 15: 844–50.
McManus C. Reversed bodies, reversed brains, and (some) reversed behaviors of zebrafish and men. Dev Cell 2005, 8: 796–7.
Logan M, Pagán-Westphal SM, Smith DM, Paganessi L, Tabin CJ. The transcription factor Pitx2 mediates situs-specific morphogenesis in response to left-right asymmetric signals. Cell 1998, 94: 307–17.
Piedra ME, Icardo JM, Albajar M, Rodriguez-Rey JC, Ros MA. Pitx2 participates in the late phase of the pathway controlling left-right asymmetry. Cell 1998, 94: 319–24.
Ryan AK, Blumberg B, Rodriguez-Esteban C, et al. Pitx2 determines left-right asymmetry of internal organs in vertebrates. Nature 1998, 394: 545–51.
Kitamura K, Miura H, Miyagawa-Tomita S, et al. Mouse PITX2 deficiency leads to anomalies of the ventral body wall, heart, extra- and periocular mesoderm and right pulmonary isomerism. Development 1999, 126: 5749–58.
Levin M, Johnson RL, Stern CD, Kuehn M, Tabin C. A molecular pathway determining left-right asymmetry in chick embryogenesis. Cell 1995, 82: 803–14.
Stern CD, Yu RT, Kakizuka A, et al. Activin and its receptors during gastrulation and the later phases of mesoderm development in the chick embryo. Dev Biol 1995, 172: 192–205.
Peeters H, Devriendt K. Human laterality disorders. Eur J Med Genet 2006, 49: 349–62.
Kosaki R, Gebbia M, Kosaki K, et al. Left-right axis malformations associated with mutations in ACVR2B, the gene for human activin receptor type IIB. Am J Med Genet 1999, 82: 70–6.
Kosaki K, Bassi MT, Kosaki M, et al. Characterization and mutation analysis of human LEFTY A and LEFTY B, homologues of murine genes implicated in left-right axis development. Am J Hum Genet 1999, 64: 712–21.
Ansley SJ, Badano JL, Blacque OE, et al. Basal body dysfunction is a likely cause of pleiotropic Bardet-Biedl syndrome. Nature 2003, 425: 628–33.
Otto EA, Schermer B, Obara T, et al. Mutations in INVS encoding inversin cause nephronophthisis type 2, linking renal cystic disease to the function of primary cilia and left-right axis determination. Nat Genet 2003, 34: 413–20.
Gebbia M, Ferrero GB, Pilia G, et al. X-linked situs abnormalities result from mutations in ZIC 3. Nat Genet 1997, 17: 305–8.
Kennedy DN, O’Craven KM, Ticho BS, Goldstein AM, Makris N, Henson JW. Structural and functional brain asymmetries in human situs inversus totalis. Neurology 1999, 53: 1260–5.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Halász, Z., Bertalan, R., Tőke, J. et al. Laterality disturbance and hypopituitarism. A case report of co-existing situs inversus totalis and combined pituitary hormone deficiency. J Endocrinol Invest 31, 74–78 (2008). https://doi.org/10.1007/BF03345570
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03345570