Abstract
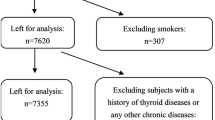
The aim of the present study is to obtain the epidemiological data on post-partum thyroiditis (PPT) firstly in Chinese women, and to try to evaluate whether excessive intake of iodine in post-partum women imposes any danger of occurring PPT. Sixty hundred and ten pregnant women were involved in the cohort just before delivery. Four hundred and eighty-eight (80%) of them accepted taking part in follow-ups more than 6 months post-partum. A blood sample was taken from participants before delivery and every 3 months post-partum for testing of serum TSH, thyroid autoantibodies. Free T3 (FT3), free T4 (FT4) and TSH receptor antibody (TRAb) were detected if TSH was abnormal. The iodine nutrition was evaluated according to the mean level of the fasting urinary iodine excretions at different times during the studying period, and participants were subgrouped into 3 categories with low, adequate and high iodine intake. For those participants who had thyroid dysfunction within 6 months post-partum, the follow-up persisted for 1 yr. Of 488 preg- nant women, PPT developed in 11.9% (58/488). Given overt and subclinical PPT, the prevalence was 7.17% (no.=35) and 4.71% (no.=23), respectively. There was a strong association between the presence of thyroid peroxidase antibody (TPOAb) at delivery and the risk of developing PPT [RR=6.76, 95% (CI) 4.42–10.34]. Overt cases had much higher titers of TPOAb than subclinical patients (all p<0.05). The median urinary iodine (MUI) of patients with PPT was significantly higher than that of healthy women (231.93 vs 199.88 μg/l p=0.00153). Both the prevalence of PPT and positive TPOAb rise with the increment of iodine intakes. Pregnant women with high iodine intake had more risk of developing PPT when compared with those with low iodine intake (RR=2.92, 95%CI 1.31–6.50). We concluded that positive TPOAb was of value for predicting the occurrence and severity of PPT, and a high iodine intake was a risk factor triggering PPT.
Similar content being viewed by others
References
Muller AF, Drexhage HA, Berghout A. Postpartum thyroiditis and autoimmune thyroiditis in women of childbearing age: recent insights and consequences for antenatal and postnatal care. Endocr Rev 2001, 22: 605–30.
Dunn JT. Editorial: Guarding our nation’s thyroid health. J Clin Endocrinol Metab 2002, 87: 486.
Dunn JT. Methods for measuring iodine in urine. Netherlands, ICCIDD, 1993.
Amino N, Mori H, Iwatani Y, et al. High prevalence of transient postpartum thyrotoxicosis and hypothyroidism. N Engl J Med 1982, 306: 849–52.
Gerstein HC. Incidence of postpartum thyroid dysfunction inpatients with type I diabetes mellitus. Ann Intern Med 1993, 118: 419–23.
Alvarez-Marfany M, Roman SH, Drexler AJ, Robertson C, Stagnaro-Green A. Long-term prospective study of postpartum thyroid dysfunction in women with insulin dependent diabetes mellitus. J Clin Endocrinol Metab 1994, 79: 10–6.
Hall R. Pregnancy and autoimmune endocrine disease. Baillieres Clin Endocrinol Metab 1995, 9: 137–55.
Lazarus JH. Prediction of postpartum thyroiditis. Eur J Endocrinol 1998, 139: 12–3.
Lazarus JH, Othman S. Thyroid disease in relation to pregnancy. Clin Endocrinol (Oxf) 1991, 34: 91–8.
Weetman AP. Prediction of post-partum thyroditis. Clin Endocrinol (Oxf) 1994, 41: 7–8.
Premawardhana LD, Parkes AB, John R, Harris B, Lazarus JH. Thyroid peroxidase antibodies in early pregnancy: utility for prediction of postpartum thyroid dysfunction and implications for screening. Thyroid 2004, 14: 610–5.
Pop VJ, de Rooy HA, Vader HL, Heide van der D, van Son MJ, Komproe I. Microsomal antibodies during gestation in relation to postpartum thyroid dysfunction and depression. Anta Endocrinol (Copenh) 1993, 129: 26–30.
Yu ZH, Liu SJ. Introduction of development of IDD in China during the past 50 years [in Chinese]. Chin J Prev Med 2003, 37: 315–8.
WHO. Assessment of Iodine deficiency disorders and monitoring their elimination: A guide for programme managers (second edition), WHO, 2001, unpublished document.
Todd CH, Allain T, Gomo ZA, Hasler JA, Ndiweni M, Oken E. Increase in thyrotoxicosis associated with iodine supplement in Zimbabwe. Lancet 1995, 346: 1563–4.
Mostbeck A, Galvan G, Bauer P, Eber O, Alifie K, Dam K. The incidence of hyperthyroidism in Austria from 1987–1995 before and after an increase in salt iodization in 1990. Eur J Nucl Med 1998, 25: 367–74.
Shan ZY, Teng WP, Li YS, et al. Comparative epidemiologic study on the prevalence of iodine-induced hypothyroidism. Chin J Endocrinol Metab 2001, 17: 71–4.
Szabolcs I, Podoba J, Feldkamp J, et al. Comparative screening for thyroid disorders in old age in areas of iodine deficiency, long-term iodine prophylaxis and abundant iodine intake. Clin Endocrinol (Oxf) 1997, 47: 87–92.
Mooij P, de Wit HJ, Drexhage HA. A high iodine intake in Wistar rats results in the development of a thyroid-associated ectopic thymic tissue and is accompanied by a low thyroid autoimmune reactivity. Immunology 1994, 81: 309.
Bagchi N, Brown TR, Urdanivia E, Sundick RS. Induction of autoimmune thyroiditis in chickens by dietary iodine. Science 1985, 230: 325–7.
Mooij P, de Wit HJ, Drexhage HA. An excess of dietary iodine accelerates the development of a thyroid-associated lymphoid tissue in autoimmune prone BB rat. Clin Immunol Immunopathol 1993, 69: 189.
Rasooly L, Burek CL, Rose NR. Iodine-induced autoimmune thyroiditis in NOD-H2h4 mice. Clin Immuunol Immunopathol 1996, 81: 287.
Othman S, Phillips DI, Lazarus JH, Parkes AB, Richards C, Hall R. Iodine metabolism in postpartum thyroiditis. Thyroid 1992, 2: 107–11.
Nøhr SB, Jorgensen A, Pedersen KM, Laurberg P. Postpartum thyroid dysfunction in pregnant thyroid peroxidase antibody positive women living in an area with mild to moderate iodine deficiency: is iodine supplementation safe? J Clin Endocrinol Metab 2000, 85: 3191–8.
Kampe O, Jansson R, Karlsson FA. Effects of L-thyroxine and iodide on the development of autoimmune postpartum thyroiditis. J Clin Endocrinol Metab 1990, 70: 1014–8.
Amino N, Tada H, Hidaka Y. Postpartum autoimmune thyroid syndrome: a model of aggravation of autoimmune disease. Thyroid 1999, 9: 705–13.
Smallridge RC, Ladenson PW. Hypothyroidism in pregnancy: consequences to neonatal health. J Clin Endocrinol Metab 2001, 86: 2349–53.
Vaquero E, Lazzarin N, De Carolis C, Valensise H, Moretti C, Romanini C. Mild thyroid abnormalities and recurrent spontaneous abortion: diagnostic and therapeutical approach. Am J Reprod Immunol 2000, 43: 204–8.
Glinoer D. Thyroid immunity, thyroid dysfunction, and the risk of miscarriage: a propos article by Vaquero et al. Mild thyroid abnormalities and recurrent spontaneous abortion: diagnostic and therapeutical approach. Am J Reprod Immunol 2000, 43: 202–3.
Fukushi M, Honma K, Fujita K. Maternal thyroid deficiency during pregnancy and subsequent neuropsychological development of the child. N Engl J Med 1999, 341: 2016.
Pop VJ, Kuijpens JL, van Baar AL, et al. Low maternal free thyroxine concentrations during early pregnancy are associated with impaired psychomotor development in infancy. Clin Endocrinol (Oxf), 1999, 50: 149–55.
Author information
Authors and Affiliations
Corresponding author
Additional information
The two authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Guan, H., Li, C., Li, Y. et al. High iodine intake is a risk factor of post-partum thyroiditis: Result of a survey from Shenyang, China. J Endocrinol Invest 28, 876–881 (2005). https://doi.org/10.1007/BF03345318
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03345318