Abstract
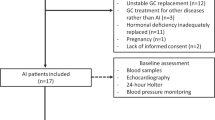
Acute myocardial infarction (AMI) is associated with a stimulation of cortisol which lasts 24 hours in patients treated by thrombolysis. Percutaneous transluminal coronary angioplasty (PTCA) is an alternative treatment for AMI which reduces the length of myocardial ischemia. Our objective was the determination of the amplitude and duration of cortisol and other hormones of the hypothalamo-pituitaryadrenal (HPA) axis release in patients undergoing PTCA. These responses were also analyzed in relation with the time of onset of AMI. The effect of coronarography with or without angioplasty in patients without AMI was also studied. Plasma ACTH, cortisol, corticotropin-releasing hormone and arginine vasopressin levels were determined during the first 48 hours in 20 patients with first AMI, treated by PTCA and in 10 patients without AMI undergoing coronarography (and angioplasty in five of them). A strong stimulation of the HPA axis was observed in AMI patients, but the duration of cortisol secretion was significantly reduced (less than 8 hours) as compared with previous studies in patients treated with thrombolysis. A clear-cut ACTH-cortisol dissociation was also observed after the third hour. ACTH and cortisol stimulation was higher in patients admitted between 04:00 h and 16:00 h than in patients admitted between 16:00 h and 04:00 h In patients without AMI, coronarography induced a moderate, but significant short-lasting ACTH and cortisol stimulation. In conclusion, our data suggest that the degree of stimulation of the HPA axis may depend upon the type of treatment and the circadian rhythm of this axis.
Similar content being viewed by others
References
Ceremuzynski L. Hormonal and metabolic reactions evoked by acute myocardial infarction. Circ. Res. 1981, 48: 767–776.
Logan R.W., Murdoch W.R. Blood levels of hydrocortisone, transaminases, and cholesterol after myocardial infarction. Lancet 1966, 2: 521–524.
Bailey R.B., Abernethy M.H., Beaven D.W. Adrenocortical response to the stress of an acute myocardial infarction. Lancet 1967, 1: 970–973.
Bain R.J.I., Fox J.P., Jagger J., Davies M.K., Littler W.A., Murray R.G. Serum cortisol levels predict infarct size and patient mortality. Int. J. Cardiol. 1992, 37: 145–150.
Wiener K. Plasma cortisol, corticosterone and urea in acute myocardial infarction: clinical and biochemical correlations. Clin. Chim. Acta 1977, 76: 243–250.
Klein A.J., Palmer L.A. Plasma cortisol in myocardial infarction: a correlation with shock and survival. Am. J. Cardiol. 1963, 11: 332–337.
Hansen B., Beck-Nielsen J., Juul J., Nielsen B.L., Nielsen F.U. Plasma hydrocortisone values in heart disease. Acta Med. Scand. 1969, 186: 411–416.
Prakash R., Parmley W.W., Horvat M., Swan H.J.C. Serum cortisol, plasma free acids, and urinary catecholamines as indicators of complications in acute myocardial infarction. Circulation 1972, 155: 736–745.
Chopra M.P., Thadani U., Aber C.P., Portal R.W., Parkes J. Plasma cortisol, urinary 17-hydroxycorticoids, and urinary vanilyl mandelic acid after acute myocardial infarction. Br. Heart. J. 1972, 34: 992–997.
Nitter-Hauge S., Kirkeby K., Alvsaker J.O., Aakwaag A. Plasma 11-hydroxycorticosteroids in acute myocardial infarction. Acta Med. Scand. 1972, 192: 535–538.
Donald R.A., Crozier I.G., Foy S.G. et al. Plasma corticotrophin releasing hormone, vasopressin, ACTH and cortisol responses to acute myocardial infarction. Clin. Endocrinol. (Oxf.) 1994, 40: 499–504.
Bain R.J.I., Poeppinghaus V.J.I., Jones G.M., Peaston M.J.T. Cortisol level predicts myocardial infarction in patients with ischaemic chest pain. Int. J. Cardiol. 1989, 25: 69–72.
Grines C.L. Primary angioplasty. The strategy of choice. N. Engl. J. Med. 1996, 335: 1313–1317.
Caraty A., Grino M., Locatelli A., et al. Insulin-induced hypoglycaemia stimulates corticotropin-releasing factor and arginine vasopressin into hypophysial portal blood of conscious unrestrained rams. J. Clin. Invest. 1990, 85: 1716–1721.
Robinson I.C.A.F. The development and evaluation of a sensitive and specific radioimmunoassay for oxytocin in unextracted plasma. Immunoassay. 1980, 1: 323–347.
Oldroyd K.G., Harvey K., Gray C.E., Beastall G.H., Cobbe S.M. β Endorphin release in patients after spontaneous and provoked acute myocardial ischaemia. Br. Heart J. 1992, 67: 230–235.
Dampney R.A.L. Functional organisation of central pathways regulating the cardiovascular system. Pharmacol. Rev. 1994, 74: 323–364.
Kiss JZ. Dynamism of chemoarchitecture in the hypothalamic paraventricular nucleus. Brain Res. Bull. 1998, 20: 966–708.
Charles C.J., Rogers S.J., Donald R.A., Ikram H., Prickett T., Richards A.M. Hypothalamo-pituitary-adrenal axis response to coronary artery embolization: an ovine model of acute myocardial infarction. J. Endocrinol. 1997, 152: 489–493.
Naito Y., Kukata J., Tamai S., et al. Biphasic changes in hypothalamo- pituitary-adrenal function during the early recovery period after major abdominal surgery. J. Clin. Endocrinol. Metab. 1991, 73: 111–117.
Roth-Isigkeit A., Schmucker P. Postoperative dissociation of blood levels of cortisol and adrenocorticotropin after coronary artery bypass grafting surgery. Steroids 1997, 62: 695–699.
Vermes I., Beishuizen A., Hampsink R.M., Haanen C. Dissociation of plasma adrenocorticotropin and cortisol levels in critically ill patients: possible role of endothelin and atrial natriuretic hormone. J. Clin. Endocrinol. Metab. 1995, 80: 1238–1242.
Charlton B.G. Adrenal cortical innervation and glucocorticoid secretion. J. Endocrinol. 1990, 126: 5–8.
Marx C., Ehrhart-Bornstein M., Scherbaum W.A., Bornstein S.R. Regulation of adrenocortical function by cytokines — relevance for immune-endocrine interaction. Horm. Metab. Res. 1998, 30: 416–420.
Guillen I., Blanes M., Gomez-Lechon M.J., Castell J.V. Cytokine signaling during myocardial infarction: sequential appearance of IL-1β and IL-6. Am. J. Physiol. 1995, 269: R229–R235.
Antoni F.A. Vasopressinergic control of pituitary adrenocorticotropin secretion comes of age. Front. Neuroendocrinol. 1993, 14: 76–122.
Zing H.H. Vasopressin and oxytocin receptors. Baillere’s Clin. Endocrinol. Metab. 1996, 10: 75–96.
Hader O., Bahr V., Hensen J., Hofbauer K.G., Oelkers WKH. Effects of a V1-vasopressin antagonist on ACTH release following vasopressin infusion or insulin-induced hypoglycemia in normal men. Acta. Endocrinol. (Copenh.) 1990, 123: 622–628.
Col J., Petein M., Van Eyll C., Cheron P., Charlier A.A., Pouleur H. Early changes in sodium and water balances in patients with acute myocardial infarction: relationship to haemodynamics and creatine kinase. Eur. J. Clin. Invest. 1984, 14: 247–254
Brar R.B., Stephanou A., Okosi A., Knight R., Lowry P.J., Latchman D.S. Regulation of the CRH promoter in ischaemic cardiac myocytes. Annual Meeting of the Endocrine Society 1998, 207 (Abst.)
Muller J.E., Stone P.H., Turi Z.G., et al. Circadian variation in the frequency of onset of acute myocardial infarction. N. Engl. J. Med. 1985, 313: 1315–1322.
Willich S.N., Linderer T., Wegscheider K., et al. Increased morning incidence of myocardial infarction in the ISAM study: absence with prior β-adrenergic blockade. Circulation. 1989, 80: 853–858.
Boden G. Hormonal and metabolic disturbances during acute and subacute myocardial infarction in man. Diabetologia 1971, 2: 240–246.
Oswald G.A., Smith C.C.T., Betteridge D.J., Yudkin J.S. Determinants and importance of stress hyperglycaemia in non-diabetic patients with myocardial infarction. Br. Med. J. 1986, 293: 917–922.
Fimognari F.P., Piccirillo G., et al. Associated daily biosynthesis of cortisol and thromboxane A2: a preliminary report. J. Lab. Clin. Med. 1996, 128: 115–121.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Paganelli, F., Frachebois, C., Velut, J.G. et al. Hypothalamo-pituitary-adrenal axis in acute myocardial infarction treated by percutaneous transluminal coronary angioplasty: Effect of time of presentation. J Endocrinol Invest 26, 407–413 (2003). https://doi.org/10.1007/BF03345195
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03345195