Abstract
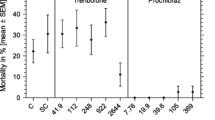
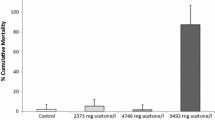
The goal of this study was to evaluate whether short 1 h sublethal assays may predict the results of 24 h lethality assays with rotifers Brachionus calyciflorus and anostracan crustaceans Thamnocephalus platyurus. The test bionts were hatched from cysts. Inhibition of ingestion was observed after 15 min of incubation of rotifers and crustaceans with the suspension of carmine and latex beads, respectively. Nine compounds with different modes of action were used as toxicants zinc ions, sodium dodecyl sulphate, p-nitrophenol, 3, 5-dichlorophenol and pharmaceuticals propranolol, fluoxetine, abamectin, doramectin and ivermectin. The toxicity values observed in the ingestion tests were very close to the mortality values over a wide range of toxicity from a low toxic surfactant to very toxic avermectins. The ratio between the 1 h EC50’s in the ingestion test and the 24 h LC50’s in the lethality test was below 2 in all cases for rotifers, and 7 in 9 cases for crustaceans. The toxicity of zinc and 3,5-dichlorophenol in the Thamnotoxkit F™ was 15-fold higher and 10 fold lower than in the ingestion test, respectively. The 24 h LC50 values are within the range of 25–400 % of the 1 h EC50 values for almost all toxicants tested with the exception of p-nitrophenol for B. calyciflorus and zinc and 3, 5-dichlorophenol for T. platyurus. Short, 1 h ingestion assays Rotoxrapid and Rapidtoxkit are good predictors of the mortality over the next 24 h and can be used as a range finding tests for representatives of pharmaceuticals and surfactants.
Similar content being viewed by others
References
ASTM, (1991). Standard guide for acute toxicity tests with the rotifer Brachionus. Annual Book of ASTM Standards, vol. 11.04, E 1440, American Society for Testing and Materials, Philadelphia, PA, USA (8 pages).
Bakoulia, P.; Karaima, C.; Rouvalis, A.; Iliopoulou-Georgudaki, J., (2008). Acute toxicity evaluation of an insecticide used in potato cultures with the use of bioassays. Fresenius Environ. Bull., 17(8A), 1002–1006 (5 pages).
Centeno, M. D. F.; Persoone, G.; Goyvaerts, M. P., (1995). Cyst-based toxicity tests. IX. The potential of Thamnocephalus platyurus as test species in comparison with Streptocephalus proboscideus (Crustacea: Branchiopoda: Anostraca). Environ. Toxicol. Water Qual., 10(4), 275–282 (8 pages).
Dahms, H. U.; Hagiwara, A.; Lee, J. S., (2011). Ecotoxicology, ecophysiology, and mechanistic studies with rotifers. Aquat. Toxicol., 101(1), 1–12 (12 pages).
De Coen, W. M.; Vangheluwe, M. L.; Janssen, C. R., (1998). The use of biomarkers in Daphnia magna toxicity testing. III. Rapid toxicity testing of pure chemicals and sediment pore waters using ingestion and digestive enzyme activity. Chemosphere, 37(13), 2677–2694 (18 pages).
Della Greca, M.; Iesce, M. R.; Isidori, M.; Montanaro, S.; Previtera, L.; Rubino, M., (2007 ). Phototransformation of amlodipine in aqueous solution: Toxicity of the drug and its photoproduct on aquatic organisms. Int. J. Photoenergy, art. no., 63459 (6 pages).
EPA, (2002). Methods for measuring the acute toxicity of effluents and receiving waters to freshwater and marine organisms. Fifth edition, US EPA Office of Water, EPA-821-R-02-012.
Fochtman, P.; Raszka, A.; Nierzedska, E., (2000). The use of conventional bioassays, microbiotests, and some “rapid” methods in the selection of an optimal test battery for the assessment of pesticides toxicity. Environ. Toxicol., 15(5), 376–384 (9 pages).
Giri, R. R.; Ozaki, H.; Ota, S.; Takanami, R.; Taniguchi, S., (2010). Degradation of common pharmaceuticals and personal care products in mixed solutions by advanced oxidation techniques. Int. J. Environ. Sci. Tech., 7(2), 251–260 (10 pages).
Hayes, K. R.; Douglas, W. S.; Fischer, J., (1996). Inter- and intra-laboratory testing of the Daphnia magna IQ toxicity test. Bull. Environ. Contam. Toxicol., 57(4), 660–666 (7 pages).
Isidori, M.; Lavorgna, M.; Nardelli, A.; Parrella, A.; Previtera, L.; Rubino, M., (2005). Ecotoxicity of naproxen and its phototransformation products. Sci. Total Environ., 348(1–3), 93–101 (9 pages).
Janssen, C. R.; Persoone, G., (1993). Rapid toxicity screening tests for aquatic biota. 1. Methodology and experiments with Daphnia magna. Environ. Toxicol. Chem., 12(4), 711–717 (7 pages).
Juchelka, C. M.; Snell, T. W., (1994). Rapid toxicity assessment using rotifer ingestion rate. Arch. Environ. Contam. Toxicol., 26(4), 549–554 (6 pages).
Juchelka, C. M.; Snell, T. W., (1995). Rapid toxicity assessment using ingestion rate of cladocerans and ciliates. Arch. Environ. Contam. Toxicol., 28(4), 508–512 (5 pages).
Kim, J. W.; Ishibashi, H.; Yamauchi, R.; Ichikawa, N.; Takao, Y.; Hirano, M.; Koga, M.; Arizono, K., (2009). Acute toxicity of pharmaceutical and personal care products on freshwater crustacean (Thamnocephalus platyurus) and fish (Oryzias latipes). J. Toxicol. Sci., 34(2), 227–232 (6 pages).
Nałęcz-Jawecki, G.; Persoone, G., (2006). Toxicity of Selected Pharmaceuticals to the Anostracan Crustacean Thamnocephalus platyurus. Comparison of Sublethal and Lethal Effect Levels with the 1h Rapidtoxkit and the 24h Thamnotoxkit Microbiotests. Environ. Sci. Poll. Res., 13(1), 22–27 (6 pages).
Nałęcz-Jawecki, G.; Sawicki, J., (1999). Spirotox-a new tool for testing the toxicity of volatile compounds. Chemosphere, 38(14), 3211–3218 (8 pages).
Palma, P.; Palma, V. L.; Fernandes, R. M.; Soares, A. M. V. M.; Barbosa, I. R., (2008). Acute toxicity of atrazine, endosulfan sulphate and chlorpyrifos to Vibrio fischeri, Thamnocephalus platyurus and Daphnia magna, relative to their concentrations in surface waters from the Alentejo region of Portugal. Bull. Environ. Contam. Toxicol., 81(5), 485–489 (5 pages).
Persoone, G., (1992). Cyst-based toxicity tests. VI: Toxkits and Fluotox tests as cost-effective tools for routine toxicity screening. Schriftenr. Ver. Wasser Boden Lufthyg. 89, 563–575 (13 pages).
Preston, B. L.; Snell, T. W.; Fields, D. M.; Weissburg, M. J., (2001). The effects of fluid motion on toxicant sensitivity of the rotifer Brachionus calyciflorus. Aquat. Toxicol., 52(2), 117–131 (15 pages).
Rapidtoxkit, (2004). Microbiotest for rapid detection of water contamination. Standard Operational Procedure. MicroBioTests Inc. Nazareth. Belgium, (19 pages). Available at: http//www.microbiotests.be
Reza, R.; Singh, G., (2010). Assessment of heavy metal contamination and its indexing approach for river water. Int. J. Environ. Sci. Tech., 7(4), 785–792 (8 pages).
Rotoxkit F™, (2000). Rotifer toxicity screening test for freshwater. Standard Operational Procedure. MicroBioTests, Nazareth, Belgium, (20 pages). Available at: http://www.microbiotests.be/
Sanchez-Fortun, S.; Barahona, M. V., (2005). Comparative study on the environmental risk induced by several pyrethroids in estuarine and freshwater invertebrate organisms. Chemosphere, 59(4), 553–559 (7 pages).
Snell, T. W., (2005). Rotifer ingestion test for rapid assessment of toxicity, in: Blaise, C.; Ferard J-F. (Eds.), Small-scale Freshwater Toxicity Investigations. Vol. 1 — Toxicity Test Methods. Springer, Dordrecht, The Netherlands. (14 pages).
Snell, T. W.; Janssen, C. R.,(1995). Rotifers in ecotoxicology: a review. Hydrobiologia, 313–314(1), 231–247 (17 pages).
Snell, T. W.; Persoone, G., (1989). Acute toxicity bioassay using rotifers. II. A freshwater test with Brachionus rubens. Aquat. Toxicol., 14(1), 81–92 (12 pages).
Snell, T. W.; Joaquim-Justo, C., (2007). Workshop on rotifers in ecotoxicology. Hydrobiologia, 593(1), 227–232 (6 pages).
Thamnotoxkit F™, (1995). Crustacean toxicity screening test for freshwater. Standard Operational Procedure. MicroBioTests Inc. Nazareth. Belgium. (23 pages). Available at: http://www.microbiotests.be/
Törökné, A.; Laszlo, E.; Chorus, I.; Fastner, J.; Heine, R.; Padisak, J.; Barbosa, F. A., (2000). Water quality monitoring by Thamnotoxkit F™ including cyanobacterial blooms. Water Sci. Tech., 42(1–2), 381–385 (5 pages).
Törökné, A.; Vasdinnyei, R.; Asztalos, B. M., (2007). A rapid microbiotest for the detection of cyanobacterial toxins. Environ. Toxicol., 22(1), 64–68 (5 pages).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jawecki, G.N., Szczęsny, Ł., Solecka, D. et al. Short ingestion tests as alternative proposal for conventional range finding assays with Thamnocephalus platyurus and Brachionus calyciflorus . Int. J. Environ. Sci. Technol. 8, 687–694 (2011). https://doi.org/10.1007/BF03326253
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03326253