Abstract
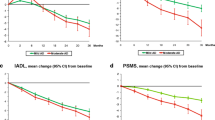
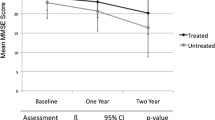
Background and aims: The most successful therapeutic approaches to Alzheimer’s disease (AD) have involved acetylcholinesterase inhibitors (ChEIs). In view of the different response rates to ChEIs therapy, it is important to identify the pharmacokinetic and pharmacodynamic mechanisms which may interfere with this effect. The aim of the study is to evaluate the efficacy on cognition of donepezil, a cholinesterase inhibitor, in a sample of mild to moderate AD patients with various serum albumin levels, a condition modifying drug distribution. Methods: Ninety-eight Alzheimer patients treated with donepezil were analyzed in an outpatient clinic between January 2003 and January 2005. At study entry, participants underwent multidimensional assessment evaluating cognitive, functional and psychobehavioral domains. All concomitant illnesses and treatments were recorded. Patients were grouped in three categories (with low, medium and high albumin levels). Results: The total sample of patients showed cognitive improvement from baseline of the ADAS Cog score at three months (ADAS Cog mean change −1.4+5.4; p=0.01), cognitive stabilization at nine (ADAS Cog mean change 0.03+6.7; p=ns), and not statistically significant worsening at fifteen months (ADAS Cog mean change 0.9+7.3; p=ns). The low serum albumin level group was associated with a greater response to donepezil. In fact, cognition, evaluated by the ADAS Cog mean change from baseline, improved during the first 15 months of treatment in the low serum albumin level group, but worsened in the two higher groups. Conclusion: Our preliminary data suggest that serum albumin level should be monitored to evaluate the clinical efficacy of ChEIs therapy.
Similar content being viewed by others
References
Racchi M, Porrello E, Lanni C, Lenzken SC, Mazzucchelli M, Govoni S. Role of acetylcholinesterase inhibitors in pharmacological regulation of amyloid precursor protein processing. Aging Clin Exp Res 2006; 18: 149–52.
Cummings JL, Kaufer D. Neuropsychiatric aspects of Alzheimer’s disease: the cholinergic hypothesis revisited. Neurology 1996; 47: 876–83.
Corey-Bloom J, Anand R, Veach J, ENA 713 B352 Study Group. A randomized trial evaluating the efficacy and safety of ENA 713 (rivastigmine tartrate), a new cholinesterase inhibitor, in patients with mild to moderately severe Alzheimer’s disease. Int J Geriatr Psychopharmacol 1998; 1: 55–65.
Pedone C, Lapane KL, Mor V, Bernabei R. Donepezil use in US nursing homes. Aging Clin Exp Res 2004; 16: 60–7.
Rogers SL, Farlow MR, Doody RS et al. A 24-week, double-blind, placebo-controlled trial of donepezil in patients with Alzheimer’s disease. Neurology 1998b; 50: 136–45.
Burns A, Rossor M, Hecker J et al. The effects of donepezil in Alzheimer’s disease — results from a multinational trial. Dement Geriat Cog Dis 1999; 10: 237–44.
Rogers SL, Doody RS, Pratt RD, Ieni JR. Long-term efficacy and safety of donepezil in the treatment of Alzheimer’s disease: final analysis of a US multicentre open-label study. Eur Neuropsychopharmacol 2000; 10: 195–203.
Bellelli G, Lucchi E, Minicuci N et al. Results of a multi-level therapeutic approach for Alzheimer’s disease subjects in the “real world” (CRONOS project): a 36-week follow-up study. Aging Clin Exp Res 2005; 17: 54–61.
Rozzini L, Vicini Chilovi B, Bellelli G et al. Effects of cholinesterase inhibitors appear greater in patients on established antihypertensive therapy. Int J Geriatr Psychiatry 2005; 20: 547–51.
Foster RH, Plosker GL. Donepezil. Pharmacoeconomic implication of therapy. Pharmacoeconomics 1999; 16: 99–114.
Lanctot KL, Herrmann N, LouLou MM. Correlates of response to acetylcholinesterase inhibitor therapy in Alzheimer’s disease. J Psychiatry Neurosci 2003; 28: 13–26.
Wood DM, Ford JM, Roberts CJ. Variability in the plasma protein binding of velnacrine (1-hydroxy tacrine hydrochloride). A potential agent for Alzheimer’s disease. Eur J Clin Pharmacol 1996; 50: 115–9.
Calderon-Margalit R, Adler B, Abramson JH et al. Butyrylcholinesterase activity, cardiovascular risk factors, and mortality in middle-aged and elderly men and women in Jerusalem. Clin Chem 2006; 52: 845–52.
McKhann G, Drachman D, Folstein M et al. Clinical diagnosis of Alzheimer’s disease. Report of the NINCDS-ADRDA work group under the auspices of the Department of Human Services Task Force on Alzheimer’s Disease. Neurology 1984; 34: 939–44.
Folstein MF, Folstein SE, McHugh PR. Mini-Mental State: A practical method for grading the cognitive state of patients for the clinician. J Psych Res 1975; 12: 189–98.
Fioravanti M, Nacca D et al. The Italian version of the Alzheimer’s disease Assessment Scale, ADAS: psychometric and normative characteristics from a normal aged population. Arch Gerontol Geriatr 1994; 19: 21–30.
Hughes CP, Berg L, Danziger WL et al. A new clinical scale for the staging of dementia. Br J Psychiatry 1982; 140: 566–72.
Lawton MP, Brody EM. Assessment of older people: self maintaining and instrumental activities of daily living. Gerontologist 1969; 9: 179–86.
Katz S, Downs TD, Cash HR, Grotz RC. Progress in development of the index of ADL. Gerontologist 1970; 10: 20–30.
Sheikh JI, Yesavage JA. Geriatric Depression Scale (GDS): Recent evidence and development of a shorter version. Clinical Gerontology: A Guide to Assessment and Intervention NY: The Haworth Press, 1986: 165–73.
Cummings JL, Mega M, Gray K et al. The Neuropsychiatric Inventory: comprehensive assessment of psychopathology in dementia. Neurology 1994; 44: 2308–14.
www.abbottdiagnostics.com/Products/Reagents_by_Condition/testdetail.cfm
Statistical Package for the Social Sciences for Windows, release 11.5.1 (Italian version). Chicago (IL): SPSS Inc: 2002.
Rozzini L. Vicini Chilovi B, Bertoletti E et al. Cognitive and psychopathologic response to rivastigmine in dementia with Lewy bodies compared to Alzheimer’s Disease: a case control study. Am J Alzheimers Dis Other Demen 2007; 22: 42–7.
Sugimoto H, Iimura Y, Yamanishi et al. Synthesis and structure-activity relationships of acetylcholinesterase inhibitors: 1-ben-zyl-4-[(5,6-dimethoxy-l-oxoindan-2-yl)methyl] piperidine hydrochloride and related compounds. J Med Chem 1995; 38: 4821–9.
www.pfizer.com/htlm/pi’s/aricepti.html
Gotti R, Bertucci C, Andrisano V et al. Study of donepezil binding to serum albumin by capillary electrophoresis and circular dichroism. Anal Bioanal Chem 2003; 377: 875–9.
Tillement JP, Lhoste F, Giudicelli JF. Diseases and drug protein binding. Clin Pharmacokinet 1978; 3: 144–54.
Morel M, Thomassin M, Berthelot A et al. Modelisation of the association mechanism of a series of huperzine derivates used for Alzheimer Disease with human serum albumin: effect of the magnesium cation. Chromatographia 2005; 61: 253–8.
Tiseo PJ, Vargas R, Perdomo CA, Friedhoff LT. An evaluation of the pharmacokinetics of donepezil HC1 in patients with impaired hepatic function. Br J Clin Pharmacol 1998; 46 (Suppl 1): 51–5.
Tiseo PJ, Foley K, Friedhoff LT. An evaluation of the pharmacokinetics of donepezil HC1 in patients with moderately to severely impaired renal function. Br J Clin Pharmacol 1998; 46 (Suppl 1): 56–60.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rozzinii, L., Chilovi, B.V., Bertoletti, E. et al. Serum albumin level interferes with the effect of Donepezil in Alzheimer’s disease. Aging Clin Exp Res 20, 509–512 (2008). https://doi.org/10.1007/BF03324877
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03324877