Abstract
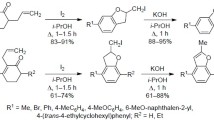
Room temperature Rap-Stoermer condensation of α-haloacetophenone with various 2-hydroxyarylaldehydes mediated by KF/Al2O3 resulted in sole formation of good to excellent yields of various substituted benzofurans in the absence solvent or extra stimulant.
Similar content being viewed by others
References
T.J. Simpson, in: R.H. Thomson (Ed.), The Chemistry of Natural Products, Blackie, London, 1985
F.M. Dean, in: J. Apsimon (Ed.), The Total Synthesis of Natural Products, Wiley, New York, vol. 1 (1973) 513.
A.R. Katritzky, Y. Ji, Y. Fang, I. Prakash, J. Org. Chem. 66 (2001) 5613
K.K. Park, H. Seo, J.-G. Kim, I.-H. Suh, Tetrahedron Lett. 41 (2000) 1393
K.C. Nicolaou, S.A. Snyder, A. Bigot, J.A. Pfefferkon, Angew. Chem. Int. Ed. 39 (2000) 1093
K.K. Park, I.K. Han, J.W. Park, J. Org. Chem. 66 (2001) 6800
A. Shafiee, E. Behnam, J. Heterocycl. Chem. 15 (1978) 589.
N. Buu-Hoi, G. Saint-Ruf, T.B. Loc, N.D. Xuong, J. Chem. Soc. (1957) 2593.
K. Benkli, N. Gundogdu-Karaburun, A.C. Karaburun, U. Ucucu, S. Demirayak, N. Kiraz, Arch. Pharm. Res. 26 (2003) 202
N. Gundogdu-Karaburun, K. Benkli, Y. Tunali, U. Ucucu, S. Demirayak, Eur. J. Med. Chem. 41 (2006) 651.
P.G. Baraldi, R. Romagnoli, I. Beria, P. Cozzi, C. Geroni, N. Mongelli, N. Bianchi, C. Mischiati, R. Gambari, J. Med. Chem. 43 (2000) 2675.
J. Li, T.S. Rush, III, W. Li, D. De Vincentis, X. Du, Y. Hu, J.R. Thomason, J.S. Xiang, J.S. Skotnicki, S. Tam, K.M. Cunningham, P.S. Chockalingam, E.A. Morris, J.I. Levin, Bioorg. Med. Chem. Lett. 15 (2005) 4961
V. Pestellini, A. Giolitti, F. Pasqui, L. Abelli, C. Cutrufo, G. De Salvia, S. Evangelista, A. Meli, Eur. Med. Chem. 23 (1988) 203.
A.Y. Leung, S. Foster, Encyclopedia of Common Natural Ingredients Used in Food, Drugs, and Cosmetics, Wiley, New York, 1996.
B. Elvers, S. Hawkins, G. Schulz, Ulmann’s Encyclopedia of Industrial Chemistry, Optical Brighteners, 5th ed., Vol A18, VCH, Weinheim (1999) 153.
S. Thielges, E. Meddah, P. Bisseret, J. Eustache, Tetrahedron Lett. 45 (2004) 907
M.V. Gil, E. Roman, J.A. Serrano, Tetrahedron Lett. 41 (2000) 10201
J.W. Herndon, Y. Zhang, H. Wang, K. Wang, Tetrahedron Lett. 41 (2000) 8687
A. Arrault, F. Touzeau, G. Guillaument, J.Y. Merour. Synthesis (1999) 1241.
D.D. Hennings, S. Iwasa, V.H. Rawal, Tetrahedron Lett. 36 (1997) 6379
X. Xie, B. Chen, J. Lu, J. Han, X. She, X. Pan, Tetrahedron Lett. 45 (2004) 6235
S.W. Youn, J.I. Eom, Org. Lett. 7 (2005) 3355.
D. Bogdal, M. Warzala, Tetrahedron 56 (2000) 8769
K.K. Park, J. Jeong. Tetrahedron 61 (2005) 545
E. Bellur, I. Freifeld, P. Langer, Tetrahedron Lett. 46 (2005) 2185
D. Bogdal, S. Bednarz, M. Lukasiewicz, Tetrahedron 62 (2006) 9440
G.A. Kraus, N. Zhang, J.G. Verkade, M. Nagarajan, P.B. Kisanga, Org. Lett. 2 (2000) 2409
M.C. Cruz, J. Tamariz, Tetrahedron Lett. 45 (2004) 2377.
W.M. Dai, K.W. Lai, Tetrahedron Lett. 43 (2002) 9377
C.G. Bates, P. Saejueng, J.M. Murphy, D. Venkataraman, Org. Lett. 4: (2002) 4727.
E. Rap, Gazz. Chim. Ital. 285 (1895) 2511
R. Stoermer, Liebigs. Ann. Chem. 312 (1900) 331.
M.L.N. Rao, D.K. Awasthi, D. Banerjee, Tetrahedron Lett. 48 (2007) 431.
K. Yoshizawa, S. Toyota, F. Toda, I. Csoregh, Green Chem. 5 (2003) 353.
J. Habermann, S.V. Ley, R. Smits. J. Chem. Soc., Perkin Trans. 1 (1999) 2421.
R.S. Varma, D. Kumar, P.J. Liesen, J. Chem. Soc., Perkin Trans. 1 (1998) 4093.
B.E. Blass, Tetrahedron, 58 (2002) 9301.
W.C. Wong, D. Wang, C. Forray, P.J.J. Vaysse, T.A. Branchek, C. Gluchowski, Bioorg. Med. Chem. Lett. 4 (1994) 2317.
V.K Yadav, K.K. Kapoor, Tetrahedron 52 (1996) 3659.
J. Yamawaki, T. Ando, Chem. Lett. (1980) 533
J. Yamawaki, T. Ando, T. Hanafusa, Chem. Lett. (1981) 1143.
F. Polyak, T. Dorofeeva, R. Sturkovich, Y. Goldberg, Synth. Commun. 21 (1991) 239.
Q.S. Hu, C.M. Hu, Chin. Chem. Lett. 8 (1997) 665.
H.M.S. Kumar, B.V.S. Reddy, E.J. Reddy, J.S. Yadav, Green Chem. 1 (1999) 141.
J. Yamawaki, T. Kawate, T. Ando, T. Hanafusa, Bull. Chem. Soc. Jpn. 56 (1983) 1885.
A.K.S.B. Rao, C.G. Rao & D.B. Singh, Synth. Commun. 21 (1991) 443.
F. Berree, E. Marchand, G. Morel. Tetrahedron Lett. 33 (1992) 6155.
B. Peschke, J.G. Bundgaard, J. Breinholt, Tetrahedron Lett. 42 (2001) 5127.
A. Sharifi, M. Mirzaei, M.R. Naimi-Jamal, J. Chem. Res. (2002) 628
A. Sharifi, M. Mirzaei, M.R. Saidi, Tetrahedron Lett. 40 (1999) 1179
A. Sharifi, M. Mirzaei, M.R. Naimi-Jamal, Monatsh. Chem. 137 (2006) 213
A. Sharifi, M. Mirzaei, M.R. Naimi-Jamal, Synth. Commun. 35 (2005) 1039
A. Sharifi, R. Salimi, M. Mirzaei, M.S. Abaee, Synth. Commun. 37 (2007) 1825.
L.A. Paquette, Encyclopedia of Reagents for Organic Synthesis, Vol 6, John Wiley & Sons, Chichester (1995) 4223.
L. Capuano, A. Ahlhelm, H. Hartmann, Chem. Ber. 119 (1986) 2069
G. Litkei, K. Gulacsi, S. Antus, Z. Dinya, Synth. Commun. 26 (1996) 3061
V.G.S. Box, P.C. Meleties, Tetrahedron Lett. 39 (1998) 7059.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Professor Dr. H. Firouzabadi on the occasions of his 65th birthday and retirement
Rights and permissions
About this article
Cite this article
Sharifi, A., Abaee, M.S., Tavakkoli, A. et al. An efficient and general procedure for room-temperature synthesis of benzofurans under solvent-free conditions using KF/Al2O3 . JICS 5 (Suppl 1), S113–S117 (2008). https://doi.org/10.1007/BF03246499
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03246499