Abstract
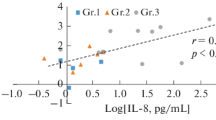
Objective: A systemic inflammatory response after open-heart surgery using cardiopulmonary bypass may be responsible for postoperative organ dysfunction. Ulinastatin, a protease inhibitor, plays an important role in host defense under periods of stress.Methods: We studied the efficacy of ulinastatin on changes in acute-phase reactive substances during and after open-heart surgery. Patients undergoing open-heart surgery were divided into an ulinastatin group (Group U) and a control group (Group C). In Group U, we introduced 600,000 units of ulinastatin into a priming solution for cardiopulmonary bypass, 300,000 units into a cardiopulmonary bypass circuit at the removal of aortic cross-clamping, and 300,000 units a day for 5 days following surgery.Results: Immediately after cardiopulmonary bypass, α1-antitrypsin levels decreased significantly in both groups, and increased significantly on the second day after surgery. Ulinastatin levels decreased after cardiopulmonary bypass in Group C. Significantly high levels of ulinastatin were obtained in Group U. Interleukin-6, interleukin-8, and polymorphonuclear elastase were markedly induced, and high levels of plasma concentration continued for several days after surgery. At all sample points, these concentrations in Group U tended to be lower than those in Group C. A significantly positive correlation was seen between the maximum levels of interleukin-8 and polymorphonuclear elastase, but these cytokine and polymorphonuclear elastase levels did not correlate with parameters such as the duration of anesthesia, surgery, cardiopulmonary bypass, or aortic cross-clamping.Conclusions: Our study suggests that high-dose ulinastatin administration to maintain a sufficient concentration of circulating protease inhibitors may suppress overinduction of cytokines and polymorphonuclear elastase in open-heart surgery.
Similar content being viewed by others
References
Miller AB, Armstrong L, Linden J, Moat N, Ekroth R, Westwick J, et al. Cytokine production and hemofiltration in children undergoing cardiopulmonary bypass. Ann Thorac Surg 1993; 56: 1499–502.
Journois D, Pouard P, Greeley WJ, Mauriat P, Vouhé P, Safran D. Hemofiltration during cardiopulmonary bypass in pediatric cardiac surgery. Effects on hemostasis, cytokines, and complement components. Anesthesiology 1994; 81: 1181–89.
Hill GE, Alonso A, Thiele GM, Robbins RA. Gluco-corticoids blunt neutrophil CD11b surface glycoprotein upregulation during cardiopulmonary bypass in humans. Anesth Analg 1994; 79: 23–7.
Jorens PG, de Jongh R, de Backer W, van Damme J, van Overveld F, Bossaert L, et al. Interkukin-8 production in patients undergoing cardiopulmonary bypass: the influence of pretreatment with methylprednisolone. Am Rev Respir Dis 1993; 148: 890–5.
Inaba H, Kochi A, Yorozu S. Suppression by methyl-prednisolone of augmented plasma endotoxin-like activity and interleukin-6 during cardiopulmonary bypass. Br J Anesth 1994; 72: 348–50.
Endo S, Inada K, Yamashita H, Takakuwa T, Nakae H, Yamada Y, et al. The inhibitory actions of protease inhibitors on the production of polymorphonuclear leukocyte elastase and interleukin 8. Res Commun Chem Pathol Pharmacol 1993; 82: 27–34.
Ishikawa A, Fukao K, Tsuji K, Osada A, Yamamoto Y, Iwasaki Y. Can ulinastatin be an effective inhibitor of human polymorphonuclear granulocyte elastase under severe stressed state? (Eng abstr). Nippon Geka Gakkai Zasshi 1993; 94: 449–55.
Doi H, Andoh T, Okutsu Y. Effects of ulinastatin on the serum activities of granulocyte elastase, β-glucuronidase and pancreatic secretory tripsin inhibitor in patients having open cardiac surgery (Eng abstr). Masui 1988; 37: 975–80.
Mitsuhata H, Enzan K, Hasegawa J, Matsumoto S, Yabe M, Matsumoto J. Effects of ulinastatin on changes of plasma granulocyte elastase, myeloperoxidase and fibronectin during and after open heart surgery (Eng abstr). Masui 1990; 39: 1164–71.
Enzan K, Mitsuhata H, Masaki Y, Matsumoto J, Shigeomi S, Komatsu H, et al. Effects of ulinastatin on granulocyte elastase and fibronectin in patients undergoing cardiopulmonary bypass (Eng abstr). Masui 1991, 40: 1625–31.
Butler J, Rocker GM, Westaby S. Inflammatory response to cardiopulmonary bypass. Ann Thorac Surg 1993; 55: 552–9.
Egami H, Arakawa H, Sakamoto K, Ikei S, Ogawa M. The role of cytokines in surgical trauma (in Japanese). Gekachiryo 1991; 65: 149–55.
Casey LC. Role of cytokines in the pathogenesis of cardiopulmonary-induced multisystem organ failure. Ann Thorac Surg 1993; 56: S92–6.
Finn A, Naik S, Klein N, Levinsky RJ, Strobel S, Elliott M. Interleukin-8 release and neutrophil degranulation after pediatric cardiopulmonary bypass. J Thorac Cardiovasc Surg 1993; 105: 234–41.
Markewitz A, Faist E, Lang S, Endres S, Hültner L, Reichart B. Regulation of acute phase response after cardiopulmonary bypass by immunomodulation. Ann Thorac Surg 1993; 55: 389–94.
Holzheimer RG, Molloy RG, Görlach H, Wilkert S, Hehrlein F. IL-6 and TNFα release in association with neutrophil activation after cardiopulmonary bypass surgery. Infection 1994; 22: 37–42.
Hennein HA, Ebba H, Rodriguez JL, Merrick SH, Keith FM, Bronstein MH, et al. Relationship of the proinflammatory cytokines to myocardial ischemia and dysfunction after uncomplicated coronary revascularization. J Thorac Cardiovasc Surg 1994; 108: 626–35.
Steinberg JB, Kapelanski DP, Olson JD, Weiler JM. Cytokine and complement levels in patients undergoing cardiopulmonary bypass. J Thorac Cardiovasc Surg 1993; 106: 1008–16.
Kalfin RE, Engelman RM, Rousou JA, Flack III JE, Deaton DW, Kreutzer DL, et al. Induction of interleukin-8 expression during cardiopulmonary bypass. Circulation 1993, 88: 401–6.
Kawamura T, Wakusawa R, Okada K, Inada S. Elevation of cytokines during open heart surgery with cardiopulmonary bypass: participation of interleukin 8 and 6 in reperfusion injury. Can J Anesth 1993; 40: 1016–21.
Frering B, Philip I, Dehoux M, Rolland C, Langlois JM, Desmonts JM. Circulating cytokines in patients undergoing normothermic cardiopulmonary bypass. J Thorac Cardiovasc Surg 1994; 108: 636–41.
Elliott MJ, Finn AHR. Interaction between neutrophils and endothelium. Ann Thorac Surg 1993; 56: 1503–8.
Moat NE, Rebuck N, Shore DF, Evans TW, Finn AHR. Humoral and cellular activation in a simulated extra-corporeal circuit. Ann Thorac Surg 1993; 56: 1509–14.
Menasché P, Haydar S, Peynet J, Du Buit C, Merval R, Bloch G, et al. A potential mechanism of vasodilation after warm heart surgery: the temperature-dependent release of cytokines. J Thorac Cardiovasc Surg 1994; 107: 293–9.
Kawai T, Wada Y, Nishiyama K, Kanki Y, Ohga K, Oka T, et al. Usefulness of ulinastatin as a radical scavenger for protection of reperfusion injury after myocardial ischemia in open heart surgery (Eng abstr). J Jpn Assn Thorac Surg 1991; 39: 2157–62.
Shimai S, Takano T, Kiuchi K, Mori N, Ibuki C, Seino Y, et al. Reduction of the extent of ischemic myocardial injury by ulinastatin in the early stage of acute myocardial infarction (in Japanese). Heart 1989; 21: 1076–81.
Yoshitomi H. Experimental studies on prospective effects of ulinastatin on “reperfusion injury” using isolated working rat heart model (in Japanese). J Nagoya City Univ Med Assn 1993; 44: 143–55.
Furunaga A, Tsuboi H, Okada H, Gohra H, Hamano K, Sugi K, et al. Clinical evaluation of patients with cardiopulmonary bypass by plasma granulocyte elastase (Eng abstr). J Jpn Assn Thorac Surg 1995; 43: 1716–19.
Miura M, Sugiura T, Aimi Y, Yasuda K, Ito S, Baba E, et al. Effects of ulinastatin on PMNL and vascular endothelial injury in patients undergoing open heart surgery with CPB (Eng abstr). Masui 1998; 47: 29–35.
Ogawa M. Systemic inflammatory response syndrome: a concept for avoiding organ dysfunction induced by a “second attack.” Surg Today 1998; 28: 679–81.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sato, Y., Ishikawa, S., Otaki, A. et al. Induction of acute-phase reactive substances during open-heart surgery and efficacy of ulinastatin. Jpn J Thorac Caridovasc Surg 48, 428–434 (2000). https://doi.org/10.1007/BF03218170
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03218170