Abstract
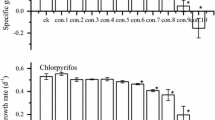
Herbicides such as atrazine, diuron, simazine and glyphosate are of growing concern with respect to ecotoxicity as they are often encountered in outdoor water samples. The present study evaluates the potential damage caused by these xenobiotics on physiology of the aquatic plantLemna sp., thereby proposing its use as a probable bioindicator. Comparative study of a panel of classical endpoints (frond number and relative growth rate) and chlorophyll fluorescence parameters (Fo, Fm, NPQ, Fv/Fm and ETRmax) revealed that diuron caused maximum inhibition of increase in frond number. This coincided with the toxicity ranking obtained on the basis of RGRarea and RGRFW i.e. diuron>atrazine>simazine>glyphosate. The chlorophyll (chl)a fluorescence parameters revealed a concentration dependent decline in maximal fluorescence (Fm) in the plants exposed to diuron, atrazine and simazine; this decline was negligible in presence of glyphosate. Besides, an EC50 of 0.009 (0.008–0.010) mg/L was recorded in case of Fv/Fm of the diuron exposedLemna sp.; furthermore, the glyphosate exposed plant demonstrated EC50s>16 mg/L. ETRmax ofLemna sp. significantly (p<0.001) declined in presence of diuron, atrazine and simazine, whereas glyphosate did not cause any significant (p>0.05) reduction. A steady concentration-dependent decline in chl a fluorescence parameters ofLemna sp. (specifically Fv/Fm and ETRmax) as compared to the classical endpoints, demonstrated their superiority and sensitivity in detection of herbicides in the aquatic bodies. This study emphasizes on the probable use of chlorophyll fluorescence ofLemna sp. as a tool or bioindicator in evaluation of herbicides.
Similar content being viewed by others
References
Relyea, R. A. The impact of insecticides and herbicides on the biodiversity and productivity of aquatic communities.Ecol. Appl. 15, 618–627 (2005).
Babu, T. S., Tripuranthakam, S. & Greenberg, B. M. Biochemical responses of the aquatic higher plantLemna gibba to a mixture of copper and 1,2-dihydroxyanthraquinone: Synergistic toxicity via reactive oxygen species.Environ. Toxicol. Chem. 24, 3030–3036 (2005).
Küster, A., Pohl, K. & Altenburger, R. A fluorescencebased bioassay for aquatic macrophytes and its suitability for effect analysis of non-photosystem II inhibitors.Env. Sci. Pol. Res. 14, 377–383 (2007).
Jones, R. The ecotoxicological effects of Photosystem II herbicides on corals.Mar. Poll. Bull. 51, 495–506 (2005).
Kida, T., Takano, S., Ishikawa, T. & Shibai, H. A simple bioassay for herbicidal substances of microbial origin by determining de novo starch synthesis in leaf segments.Agric. Biol. Chem. 49, 1299–1303 (1985).
Hulsen, K., Minne, V., Lootens, P., Vandecasteele, P. & Hofte M. A chlorophylla fluorescence-basedLemna minor bioassay to monitor microbial degradation of nanomolar to micromolar concentrations of linuron.Environ. Microbiol. 4, 327–337 (2002).
Lazar, D. & Naus, J. Statistical properties of chlorophyll fluorescence induction parameters.Photosynthetica.35, 121–128 (1998).
Philippe, J., Baosheng, Q. & Charles, P. D. Use of chlorophyll fluorescence as a tool for determination of herbicide toxic effect: Review.Toxicol. Environ. Chem. 89, 609–625 (2007).
Endo, R. & Omasa, K. Chlorophyll fluorescence imaging of individual algal cells: effects of herbicide onSpirogyra distenta at different growth stages.Environ. Sci. Technol. 38, 4165–4168 (2004).
Brain, R. S. & Solomon, K. R. A protocol for conducting 7-day daily renewal tests withLemna gibba.Nat. Protoc. 2, 979–987 (2007).
Claudia, S., Simon, M., Spranger, J. & Baumgartner, S. Test system stability and natural variability of aLemna gibba L.Bioassay PLoS ONE 3, e3133. doi: 10.1371/journal.pone.0003133 (2008).
Schreiber, U. inPulse-Amplitude-Modulation (PAM) Fluorometry and Saturation Pulse Method: an Overview, Chlorophyll a Fluorescence: A Signature of Photosynthesis (eds Papageorgiou, G. C., Govindjee) 279–319 (Springer, Amsterdam, 2004).
Platt, T., Gallegosc, L. & Harrison, G. Photoinhibition of photosynthesis in natural assemblages of marine phytoplankton.J. Mar. Res. 38, 687–701 (1980).
Sobrero, M. C., Rimoldi, F. & Ronco, A. E. Effects of the glyphosate active ingredient and a formulation onLemna gibba L. at different exposure levels and assessment end-points.Bull. Environ. Contam. Toxicol. 79, 537–543 (2007).
Frankart, C., Eullaffroy, P. & Vernet G. Comparative effects of four herbicides on non-photochemical fluorescence quenching inLemna minor.Environ. Exp. Bot. 49, 159–168 (2003).
Kovach, J., Petzoldt, C., Degnil, J. & Tette, J. A method to measure the environmental impact of pesticides.New York’s Food Life Sci. Bull. 139: 1–8 (1992).
Vass, I., Turcsányi, E., Touloupakis, E., Ghanotakis, D. & Petrouleas, V. The mechanism of UV-A radiation-induced inhibition of photosystem II electron transport studied by EPR and chlorophyll fluorescence.Biochem. 41, 10200–10208 (2002).
Ralph, P. J. Herbicide toxicity ofHalophila ovalis assessed by chlorophylla fluorescence.Aqua. Bot. 66, 141–152 (2000).
Mitsou, K.et al. Growth rate effects, responses of antioxidant enzymes and metabolic fate of the herbicide Propanil in the aquatic plantLemna minor.Chemosphere 62, 275–284 (2006).
Fairchild, J. F., Ruessler, D. S., Haverland, P. S. & Carlson A. R. Comparative sensitivity ofSelenastrum capricornutum andLemna minor to sixteen herbicides.Arch. Environ. Contam. Toxicol. 32, 353–357 (1997).
Mazzeo, N., Dardano, B. & Marticornea, A. Interclonal variaiton in response to simazine stress inLemna gibba (Lemnaceae).Ecotoxicol. 7, 151–160 (1998).
Teisseire, H., Couderchet, M. & Vernet, G. Phytotoxicity of diuron alone and in combination with copper or folpet on duckweed (Lemna minor).Environ. Poll. 106, 39–45 (1999).
Michel, A., Johnson, R. D., Duke, S. O. & Scheffler, B. E. Dose-response relationships between herbicides with different modes of action and growth ofLemna paucicostata: An improved ecotoxicological method.Environ. Toxicol. Chem. 23, 1074–1079 (2004).
Marwood, C. A., Solomon K. R. & Bruce, M. G. Chlorophyll fluorescence as a bioindicator of effects on growth in aquatic macrophytes from mixtures of poly-cyclic aromatic hydrocarbons.Environ. Toxicol. Chem. 20, 890–898 (2001).
Robert, W. G., George, D. & Bruce, M. G. Using chlorophylla fluorescence to detect the onset of anthracene photoinduced toxicity inLemna gibba, and the mitigating effects of a commercial humic acid.Limnol. Oceanog. 44(3, Part 2), 878–888 (1999).
Conrad, R.et al. Changes in yield in-vivo fluorescence of chlorophyll as a tool for selective herbicide monitoring.J. Appl. Phycol. 5, 505–516 (1993).
Amrhein, N., Deus, B., Gehrke, P. & Steinruken, H. C. The site of the inhibition of the shikimate pathway by glyphosate.Plant Physiol. 66, 830–834 (1980).
Macedo, R. S., Lombardi, A. T., Omachi, C. Y. & Rorig, L. R. Effects of the herbicide bentazon on growth and photosystem II maximum quantum yield of the marine diatomSkeletonema costatum.Toxicol. in Vitro 22, 716–722 (2008).
Magnusson, M., Heimann, K. & Negri, A. P. Comparative effects of herbicides on photosynthesis and growth of tropical estuarine microalgae.Mar. Poll. Bull. 56, 1545–1552 (2008).
Muller, R.et al. Rapid exposure assessment of PSII herbicides in surface water using a novel chlorophyll a fluorescence imaging assay.Sci. Total Env. 401, 51–59 (2008).
Jones, R. J. & Kerswell, A. P. Phytotoxicity of photosystem II (PSII) herbicides to coral.Mar. Ecol. Prog. Ser. 261, 149–159 (2003).
Gausman, M. A comparison of duckweed and standard algal phytotoxicity tests as indicators of aquatic toxicology. A practicum submitted to Miami University (Oxford, Ohio), 1–45 (2006).
Casida, J. E. Pest toxicology: the primary mechanisms of pesticide action.Chem. Res. Toxicol. 22, 609–619 (2009).
Hamilton, D. J.et al. Regulatory limits for pesticide residues in water.Pure Appl. Chem. 75, 1123–1155 (2003).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumar, K.S., Han, T. Physiological response ofLemna species to herbicides and its probable use in toxicity testing. Toxicol. Environ. Health. Sci. 2, 39–49 (2010). https://doi.org/10.1007/BF03216512
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03216512