Summary
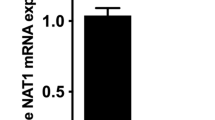
The N-acetylation of arylamines and hydrazines used as drugs may alter their pharmacological or toxicological activity. Arylamine N-acetyltransferases (NATs) are involved in drug metabolism, as they catalyse the N-acetylation of arylamine and mono-substituted hydrazine substrates. Placental metabolism regulates the nature of the chemicals which reach the developing fetus. The study of drug metabolism during pregnancy is important in determining the effect on the fetus of drugs administered to the mother and the maternal drug dose required, important if the treatment is to be effective. There are two forms of NAT in humans, NAT1 and NAT2, which are encoded at multi-allelic loci. There is inter-individual variation in both NAT1 and NAT2 activity, which has implications in drug dosage. Using a combination of enzyme activity measurements and Western blotting, this study has characterised the arylamine N-acetylation capabilities of placenta and cord blood. NAT1 activity in placenta and cord blood demonstrated inter-individual variation and the variation was in the range expected for adult NAT1 activity. The genotypes of bothNAT1 * andNAT2 * were determined using DNA prepared using placental blood clots (maternal DNA) and cord blood (fetal DNA). The results indicate that placental NAT activity is an important factor when consideringN-acetylation during pregnancy.
Similar content being viewed by others
References
Weber W.W., Hein D.W. (1985): N-acetylation pharmacogenetics. Pharmacol. Rev., 37, 25–79.
Page E.W. (1957): Transfer of materials across the human placenta. Am. J. Obstet. Gynaecol., 74, 705–718.
Simone C., Derewlany L.O., Koren G.K. (1994): Drug transfer across the placenta. Fetal Drug Ther. 21, 463–481.
Vatsis K.P., Weber W.W. (1993): Structural heterogeneity of Caucasian N-acetyltransferase at the NAT1 gene locus. Arch. Biochem. Biophys., 301, 71–76.
Blum M. Derrierre A., Grant D.M., Heim M., Meyer U.A. (1991): Molecular mechanisms of slow acetylation of drugs and carcinogens in humans. Proc. Natl. Acad. Sci. USA, 88, 5237–5241.
Evans D.A.P. (1989): N-acetyltransferase. Pharmacol. Ther., 42, 157–234.
Grant D.M., Hughes N.C., Janezic S.A. et al. (1997): Human acetyltransferase polymorphisms. Mutat. Res., 376, 61–70.
Payton M., Sim E. (1997): Genotyping human arylamine N-acetyltransferase type-1 (NAT1): the identification of a novel mutation. Pharmacogenetics, 54. In press.
Ohsako S., Deguchi T. (1990): Cloning and expression of cDNAs for polymorphic and monomorphic arylamine N-acetyltransferase from human liver. J. Biol. Chem., 265, 4630–4634.
Ilett K.F., Ingram D.M., Carpenter D.S. et al. (1994): Expression of monomorphic and polymorphic N-acetyltransferases in human colon. Biochim. Pharmacol., 47, 914–917.
Ward A., Hickman D., Gordon J.W., Sim E. (1992): Arylamine N-acetyltransferases in human colon. Biochem. Pharmacol., 44, 1099–1104.
McQueen C.A., Weber W.W. (1980): Characterization of human lymphocyte N-acetyltransferase and its relationship to isoniazid acetylator polymorphism. Biochem. Genet., 18, 889–903.
Badawi A.F., Hirvonen A., Bell D.A., Lang N.P., Kadlubar F.F. (1995): Role of aromatic amine acetyltransferases, NAT1 and NAT2, in carcinogen-DNA adduct formation in the human urinary bladder. Cancer Res., 55, 5230–5237.
Jenne J.W., Orsen M. (1965): Partial purification and properties of the isoniazid transacetylase in human liver. Its relationship to the acetylation ofpara-aminosalicylic acid in man. J. Clin. Invest., 44, 1992–2002.
Adams D.J., Seilman S., Amelizad Z., Oesch F., Wolf C.J. (1985): Identification of human cytochromes P450 analogous to form induced by phenobarbitol and 3 methylchotanthrene in the rat. Biochem. J. 232, 869–876.
Bradford M.M. (1976): a rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem., 72, 248–254.
Stanley L.A., Coroneos E., Cuff R., Hickman D., Ward A., Sim E. (1996): Immunochemical detection of arylamine N-acetyltransferases in normal and neoplastic bladder. J. Histochem. Cytochem., 44, 1059–1067.
Henderson C.J., Wolf C.R. (1992): Immunodetection of proteins by Western blotting. In: Manson M.M. (ed.) Immunochemical Protocols. Totowa, NJ: Humana Press, 221–233.
Dilella A.G., Woo S.L.C. (1987): Cloning large segments of genomic DNA using cosmid vectors. Methods Enzymol., 152, 199–212.
Hickman D., Risch A., Camilleri J.P., Sim E. (1992): Genotyping human polymorphic arylamine N-acetyltransferase. Identification of new slow allotypic variants. Pharmacogenetics, 2, 217–2613.
Cribb A.E., Grant D.M., Miller M.A., Spielburg S.P. (1991): Expression of monomorphic arylamine N-acetyltransferase (NAT1) in human leukocytes. J. Pharmacol. Exp. Ther., 259, 1241–1246.
Risch A., Smelt V., Lane D. et al. (1996): Arylamine N-acetyltransferase in erythrocytes of cystic fibrosis patients. Pharmacol. Toxicol., 78, 235–240.
Hakkola J., Pasanen M., Hukkanen J. et al. (1996): Expression of xenobiotic metabolizing cytochrome P450 forms in human full term placenta. Biochem. Pharmacol., 51, 403–411.
Pacifici G.M., Rane A. (1982): Distribution of UDP-glucuronyl transferase in different human fetal tissues. Br. J. Clin. Pharmcol., 13, 732–735.
Ward A (1995): Human arylamine N-acetyltransferase Type 1. D. Phil. Thesis, University of Oxford.
Pacifici G.M., Bencini C., Rane A. (1986): Acetyltransferases in humans: development and tissue distribution: Pharmacology, 32, 283–291.
Clemente C. Anatomy, 4th Edn. London: Williams and Wilkins.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Smelt, V.A., Mardon, H.J., Redman, C.W.G. et al. Acetylation of arylamines by the placenta. Eur. J. Drug Metab. Pharmacokinet. 22, 403–408 (1997). https://doi.org/10.1007/BF03190977
Issue Date:
DOI: https://doi.org/10.1007/BF03190977