Summary
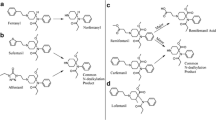
RWJ-37874, an analogue of aroyl(aminoacyl)pyrrole, is a new analgesic agent. The in vitro metabolism of RWJ-37874 was conducted using rat and human hepatic S9 in the presence of an NADPH generating systme, and API-ionspray-MS and MS/MS techniques for metabolite profiling and identification. Unchanged RWJ-37874 (66 & 86% of the sample in rat & human, respectively) plus four metabolites were profiled and tentatively identified on the basis of MS data. RWJ-37874 metabolites were formed via the following two metabolic pathways: 1. oxidative N-deethylation, and 2. pyrrole-oxidation. Pathway 1 produced a mayor and a minor metabolites, N-desethyl-RWJ-37874 (M1; 34% in rat; 13% in human) and N,N-didesethyl-RWJ-37874 (M3; <0.5% in both species), respectively. Pathway 2 formed hydroxypyrrole-RWJ-37874 (M2; <0.5% in all species), and in conjunction with step 1, formed hydroxy-M1 (M4; <0.5% in rat). RWJ-37874 is substantially metabolized in rat and human hepatic S9 fractions. However, rat appears to metabolize RWJ-37874 more extensively than human via N-dealkylation forming N-desethyl-RWJ-37874 as a major metabolite.
Similar content being viewed by others
References
Carson, J. R., Carmosin, R. J., Pitis, P. M., Vaught, J. L., Almond, H. R., Stables, J. P., Wolf, H. H., Swinyard, and White, H. S., (1997): Aroyl(aminoacyl)pyrroles, a new class of anticonvulsant agents. J. Med. Chem., 40(11), 1578–1584.
Carmosin, R. J., Carson, J. R., and Pitis, P. M., (1994): Preparation of anticonvulsant aroyl(aminoacyl)pyrroles, US Patent No. 5332736.
Carson, J. R., Pitis, P. M., and Rogers, K. E., (2000): preparation of aminoacyl(aroyl)pyrroles for treatment of neuropathic pain, US Patent No. 99120477.
Estrada, E., and Pena, A., (2000): In silico studies for the rational discovery of anticonvulsant compounds. Bioorg. Med. Chem., 8(12), 2755–2770.
Chong, M. S., and Smith, T. E., (2000): Anticonvulsants for the management of pain. Pain Reviews, 7, 129–149.
Sumner, D. D., Dayton, P. G., Cucinell, S. A., and Plostnieks, J. (1975): Metabolism of tolmetin in rat, monkey, and man, Drug Metabolism and Disposition, 3(4): 283–286.
Grindell, J. M., O’Neill, P. J., Yorgey, K. A., Schwartz, M. H., McKown, L. A., Migdalof, B. H., and Wu, W. N., (1980): The metabolism of zomepira. I. Disposition in laboratory animals and man. Drug Metabolism and Disposition, 8(3), 343–348.
Wu, W. N., Weaner, L. E., Kalbron, J., O’Neill, P. J., and Grindell, J. M., (1980): The metabolism of zomepirac sodium II. Isolation and identification of the urinary metabolites in rat, mouse, rhesus monkey, and human. Drug Metabolism and Disposition, 8(3), 349–352.
Hasegawa, J., Smith, P. C., and Benet, L. Z. (1982): Apparent intramolecular acyl migration of zomepirac glucuronide. Drug Metabolism and Disposition, 10(5), 469–473.
Wu, W. N., McKown, L. A., and J. R. Carson, (2000): In vitro metabolism of the analgesic agent, RWJ-37874, in rat and human hepatic S9 fractions (Abstract # 195. The 13th International Symposium on Microsomes and Drug Oxidation).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wu, W.N., McKown, L.A. & Carson, J.R. In Vitro metabolism of the analgesic agent, RWJ-37874, in rat and human. Eur. J. Drug Metab. Pharmacokinet. 28, 101–105 (2003). https://doi.org/10.1007/BF03190496
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03190496