Summary
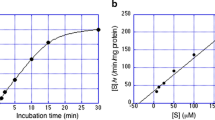
Metabolism of simvastatin (SV), a new cholesterol-lowering agent, by hepatic microsomes from male and female rats was investigated. After incubation of [14C]-SV with hepatic microsomes, radioactive metabolites were detected by HPLC. The main metabolite was 3′α-hydroxy-SV in male rats and the hydroxy open acid form of SV (SVA) in females. The 3″-hydroxy-SV and 3′, 3″-dihydroxy-SV which were observed in male rats were hardly detected in females. Specific activity for the metabolism of SV in male rats (3.97 nmol/mg protein/min) was about 9-times higher than that in females. Metabolic activity of hepatic microsomes in male rats was essentially unchanged with increase in age, whereas that in females decreased age-dependently and was very low or negligible after 7 weeks of age. Formation of 3″-hydroxy-SV and 3′, 3″-dihydroxy-SV in male rats was markedly increased with age, and that in females was negligible at all ages examined.
Similar content being viewed by others
References
Albert A.W., Chen J., Huff J., Hunt V., Kuron G. (1986): Comparative studies on the hydroxymethylglutaryl coenzyme A reductase inhibitors mevinolin, MK-733 and CS-514. IXth International Symposium on Drugs Affecting Lipid Metabolism. Abstract 8.
Olsson A.G., Molgaard J., Von Schenk H. (1986): Synvinolin in hypercholesterolemia. Lancet, 2, 390.
Mol M. J., Erkelens D.W., Gevers Leuven J.A., Schouten J. A., Stalenhoef F.H. (1986): Effects of synvinolin (MK-733) on plasma lipids in familial hypercholesterolemia. Lancet, 2, 936–939.
Stalenhoef A.F., Mol M.J., Erkelens D.W., Gevers Leuven J.A., Schouten J.A. (1986): Effect of synvinolin a new cholesterol synthesis inhibitor in familial hypercholesterolemia. Atherosclerosis, 6, 550a.
Hoffman W.F., Albert A.W., Anderson P.S., Chen J.S., Smith R.L., Willard A.K. (1986): 3-hydroxy-3-methylglutarylcoenzyme A reductase inhibitors. 4. Side chain ester derivatives of mevinolin. J. Med. Chem., 29, 849–852.
Vickers S., Duncan C.A., Rosegay A., Chen I-Wu., Duggan D.E. (1990): Metabolic disposition studies on simvastatin, a cholesterol lowering product. Drug Metab. Dispos., 18, 138–146.
Uchiyama N., Saito Y., Kagami Y., et al. (1990): Metabolic fate of simvastatin, an inhibitor of HMG-Co A reductase (1): absorption, distribution, metabolism and excretion of [14C]-simvastatin after single administration in male rats. Xenobiot. Metab. Dispos., 5, 133–149.
Ohtawa M., Uchiyama N., Saito K., Esumi Y., Jin Y. (1990): Metabolic fate of simvastatin, an inhibitor of HMG-Co A reductase (2): absorption, distribution and excretion after consecutive oral administration, and foeto-placental transfer and excretion into milk of [14C]-simvastatin in rats. Xenobiot. Metab. Dispos., 5, 151–163.
Vickers S., Duncan C.A., Vyas K.P., et al. (1990): In vitro and in vivo biotransformation of simvastatin, an inhibitor of HMG-Co A reductase. Drug Metab. Dispos., 18, 476–483.
Vyas K.P., Kari P.H., Pitzenberger S.M. (1990): Regioselectivity and stereoselectivity in the metabolism of HMG-Co A reductase inhibitors. Biochem. Biophys. Res. Commun., 166, 1155–1162.
Uchiyama N., Kagami Y., Ohtawa M. (1990): Studies on metabolic fate of simvastatin. III. Sex differences in metabolites after single administration to rats. Iyakuhin Kenkyu, 21, 1203–1214.
Takayama F., Saito K., Ohtawa M. (1991): Sex difference in the stereoselective metabolism of a new dihydropyridine calcium antagonist in the rats studied in vivo and in vitro. Xenobiotica, 21, In press.
Lowry O.H, Rosebrough N.J., Farr A.L, Randall R.J. (1951): Protein measurement with the folin phenol reagent. J. Biol. Chem., 193, 265–275.
Ryan D.E., Iida S., Wood A.W. Thomas P.E., Lieber C.S., Levin W. (1984): Characterisation of three highly purified cytochrome P-450 from hepatic microsomes of adult male rats. J. Biol. Chem., 259, 1239–1250.
Waxman D.J. (1984): Rat hepatic cytochrome P-450 isoenzyme 2c. Identification as a male-specific, developmentally induced steroid 16 alpha-hydroxylase and comparison to a female-specific cytochrome P-450 isoenzyme. J. Biol. Chem., 259, 15481–15490.
Kamataki T., Maeda K., Yamazoe Y., Nagai T., Kato R. (1983): Sex difference of cytochrome P-450 in the rat purification, characterisation and quantitation of constitutive forms of cytochrome P-450 from liver microsomes of male and female rats. Arch. Biochem. Biophys., 225, 758–770.
MacGeoch C., Morgan E.T., Halpert J., Gustafsson J.A. (1984): Purification, characterization, and pituitary regulation of the sex-specific cytochrome P-450 15 beta-hydrolase from liver microsomes of untreated female rats. J. Biol. Chem., 259, 15433–15439.
Vlasuk G.P., Walz F.G. (1982): Liver microsomal polypeptides from Fischer-344 rats affected by age, sex, and xenobiotic induction. Arch. Biochem. Biophys., 224, 248–259.
Maeda K., Kamataki T., Nagai T., Kato R. (1984): Postnatal development of constitutive forms of cytochrome P-450 in liver microsomes of male and female rats. Biochem. Pharmacol., 33, 509–512.
Waxman D.J., Dannan G.A., Guengerich F.P. (1985): Regulation of rat hepatic cytochrome P-450: age-dependent expression, hormonal imprinting and xenobiotic inducibility of sex-specific isoenzymes. Biochemistry, 24, 4409–4417.
Kamataki T., Maeda M., Shimada M., Kato R. (1986): Effect of phenobarbital, 3-methylcholanthrene and polychlorinated biphenyls on sex-specific forms of cytochrome P-450 in liver microsomes of rats. J. Biochem., 99, 841–845.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ohtawa, M., Uchiyama, N. Sex difference in metabolism of simvastatin by rat hepatic microsomes. European Journal of Drug Metabolism and Pharmacokinetics 17, 175–181 (1992). https://doi.org/10.1007/BF03190142
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03190142