Summary
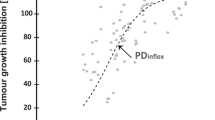
A new imidazole derivative, DP-1904, produces a selective, potent and long-acting inhibition of thromboxane A2 (TXA2) syntheses and platelet aggregation. This study was designed to investigate the pharmacokinetics and pharmacodynamics (PK/PD) of DP-1904. DP-1904 disappeared from plasma with a half-life of 20 min after i.v. dosing, and the bioavailability after oral dosing was approximately 70%. The level of serum TXB2, which is a pharmacological marker for thromboxane synthetase inhibition, was measured to characterize the pharmacodynamics of DP-1904. A marked reduction of serum TXB2 was exhibited with 1 h after both i.v. and oral doses, reflecting the rapid onset of action of DP-1904. Serum TXB2 returned to the basal level much more slowly after oral dosing than after i.v. dosing, due to the longer half-life after oral dosing. An Emax model was employed to fit the pharmacological data after oral dosing, and IC50 and Emax values were estimated to be 5.0 ng/ml and 81%, respectively. In order to test its predictability, the PK/PD model was then used to predict a pharmacological profile after i.v. dosing; good agreement between the observed and predicted values was achieved. Thus, the present modelling procedure may be useful for optimizing the therapeutic regiment of DP-1904.
Similar content being viewed by others
References
Kanao M., Watanabe Y., Kimura Y. et al. (1989): Thromboxane A2 synthetase inhibitor. 2. Synthetes and activities of tetrahydronaphthalene and indan derivatives. J. Med. Chem., 32, 1326–1334.
Toda I., Nozaki A., Kawakubo K. et al. (1990): Effects of thromboxane A2 synthetase inhibitor on ventricular fibrillation threshold during coronary artery occlusion and reperfusion. Jpn. Heart J., 31, 87–97.
Shimizu K., Kobayashi J., Iwanaga T., Kuratomi Y., Kitamura S. (1989): Effects of thromboxane A2 synthetase inhibitor, DP-1904, on the action of vasoactive substances in rabbit blood vessel smooth muscle preparations. Jpn. J. Thor. Dis., 27, 591–596.
Takami M., Takata Y., Matsumoto K., Ono S., Tsukada W. (1991): Effect of DP-1904, a new thromboxane A2 synthetase inhibitor, on guinea pig experimental asthma, Ann. N.Y. Acad. Sci., 629, 407–409.
Masumura H., Kunitada S., Irie K., Ashida S., Abe Y. (1991): A thromboxane A2 synthetase inhibitor, DP-1904, prevents rat renal injury, Eur. J. Pharmacol., 193, 321–330.
Tanaka M., Ono K., Takegoshi T. et al. (1989): The pharmacokinetics and pharmacodynamics of a new thromboxane synthetase inhibitor, 6-(1-imidazolylmethyl)-5,6,7,8-tetrahydronaphthalene-2-carboxylic acid (DP-1904), in man after single oral administration. J. Pharm. Pharmacol., 41, 680–684.
Tanaka M., Ono K., Hakusui H. et al. (1990): The pharmacokinetics and pharmacodynamics of a new thromboxane synthetase inhibitor, 6-(1-imidazolylmethyl)-5,6,7,8-tetrahydronaphthalene-2-carboxylic acid (DP-1904) in man after repeated oral doses. J. Pharm. Pharmacol., 42, 491–495.
Iwata T., Mikashima H., Takamatsu R. (1990): Correlation between pharmacokinetics and pharmacologic effects of a new imidazole thromboxane synthetase inhibitor. J. Pharm. Sci., 79, 295–300.
Nagashima R., O’Reilly RA., Levy G. (1969): Kinetics of pharmacologic effects in man: The anticoagulant action of warfarin. Clin. Pharmacol. Ther., 10, 22–35.
Barrett J.S., Gould R.J., Ellis J.D. et al. (1994): Pharmacokinetics and pharmacodynamics of L-703,014, a potent fibrinogen receptor antagonist, after intravenous and oral administration in dog. Pharm. Res., 11, 426–431.
Averbuch M., Weingeraub M., Liao J.C., Brazell R.K., Dobbs R.E. (1988): Red blood cell sorbitol lowering effects and tolerance of single doses of AL 1576 (HOE 843) in diabetic patients. J. Clin. Pharmacol., 28, 757–761.
Tanaka M., Ono K., Takegoshi T. (1988): Determination of the thromboxane synthetase inhibitor 6-(1-imidazolylmethyl)-5,6,7,8-tetrahydro-naphthalene-2-carboxy lic acid (DP-1904) in human plasma and urine using solid-phase extraction and high-performance liquid chromatography. J. Chromatogr., 426, 111–119.
Yamaoka K., Nakagawa T., Uno T. (1978): Statistical moments in pharmacokinetics. J. Pharmacokinet. Biopharm., 6, 547–558.
Yamaoka K., Tanigawara Y., Nakagawa T., Uno T. (1981): A pharmacokinetic analysis program (MULTI) for microcomputer. J. Pharmacobiodyn., 4, 879–885.
Sato, H.: Pharmaceutical Research and Development (2nd Ser.), Tanaka H., Yamaoka K. (eds) Tokyo: Hirokawa Publishing Co., 1991: 53–80.
Dayneka N.L., Garg V., Jusko W.J. (1993): Comparison of four basic models of indirect pharmacodynamic responses. J. Pharmacokinet. Biopharm., 21, 457–478.
Moskowitz M.A., Coughlin S.R. (1981): Basic properties of the prostaglandins. Stroke, 12, 696–701.
Zheng N.X., Sato H., Adachi I., Kanamoto I., Horikoshi I. (1995): Pharmacokinetic and pharmacodynamic studies of a thromboxane synthetase inhibitor, ozagrel, in rabbits. Biol. Pharm. Bull., 18, 1738–1743.
Shimizu K., Kobayashi J., Iwanaga T., Kuratomi Y., Kitamura S. (1989): Effects of thromboxane A2 synthetase inhibitor, DP-1904 on the action of vasoactive substances in guinea pig trachea and lung tissue strips. Jpn. J. Thor. Dis., 27, 339–344.
Zheng N.X., Sato H., Adachi I., Kanamoto I., Horikoshi I. (1995): Pharmacokinetic and pharmacodynamic modeling of a novel thromboxane synthetase inhibitor, DP-1904, in human. Xenobiotic Metab.Dispos., 10, 538–546.
Ramsay M.W., Grass G.M., Vallner J.J. (1991): Pharmacokinetic simulations using STELLA®: prediction of in vivo performance of oral dosage forms. Eur. J. Pharm. Biopharm., 37, 192–197.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zheng, N.X., Sato, H., Adachi, I. et al. Pharmacokinetic-pharmacodynamic modelling of DP-1904, a novel thromboxane synthetase inhibitor in rabbits, based on an indirect response model. European Journal of Drug Metabolism and Pharmacokinetics 21, 285–293 (1996). https://doi.org/10.1007/BF03189729
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03189729