Abstract
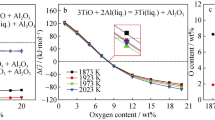
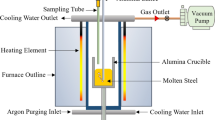
The effects of the oxygen/argon mixing ratio and temperature on the decarburization behaviour of high-carbon ferromanganese melts were investigated. The decarburization reaction was promoted and the oxidation reaction of manganese diminished by increasing temperature and decreasing the oxygen/argon mixing ratio. However, the carbon content in the melt appeared to increase during blowing at low temperatures and high oxygen/ argon mixing ratios because of faster manganese oxidation. An empirical equation was derived to predict the utilization ratio of oxygen for decarburization. The lower oxygen/argon mixing ratio and higher melt temperature were found to be essential in enhancing the efficiency of decarburization and suppressing the oxidation of manganese in the early stage of oxygen/argon refining of high-carbon ferromanganese melts.
Similar content being viewed by others
References
E. Schiirmann A. Ender, E. Höffken, H. Litterscheld and C. H. Schütz,Stahl Eisen 113, 77 (1993).
B. D. You,J. Kor. Inst. Met. & Mater. 33, 1508 (1995).
D. S. Kozak and L. R. Matricardi,Iron & Steelmaker 4, 28 (1981).
R. G. Ward,J. Uron & Steel Inst. 201, 11 (1963).
W. Dresler,Canad.+ Met. Quarter 28, 109 (1989).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
You, BD., Park, KY., Pak, JJ. et al. Oxygen refining of molten high-carbon ferromanganese. Metals and Materials 5, 395–399 (1999). https://doi.org/10.1007/BF03187764
Issue Date:
DOI: https://doi.org/10.1007/BF03187764