Abstract
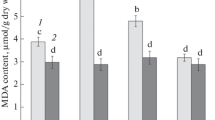
Superoxide dismutase (SOD) activity in the leaves of transgenic tobacco plants with betaine aldehyde dehydrogenase (BADH) gene was about 36% higher than that in the control plants (parent plants), activities of peroxidase (POD) and catalase (Cat) increased by about 62% and 88% respectively. Activities of ascorbate peroxidase (AsSPOD), dehydroascorbate redutase (DAsAR) and glutathione reductase (GR) in ascorbate-glutothion pathway located at chloroplasts increased by 67.7%, 47.9% and 38.8% respectively. These results indicated that the H2O2 produced by SOD catalyzing superoxide anion radicals (O2 −) could be fully decomposed, and could not derive to form the strongest toxicant radicals · OH. This is the first report to elucidate quantitatively that the activities of two kinds of antioxidative enzymes decomposed radicals and active oxygen were matched. Photoinhibition tolerant capacity of the transgenic tobacco plants was 35% higher than that in the parent plants. Increment of photoinhibition tolerant capacity in the transgenic tobacco plants might be due to increment of antioxidative enzymes activities, in turn being able to more effectively scavenge active oxygen and radicals, protect organization and function of chloroplasts. These results showed that the increment of antioxidative enzymes activities in the transgenic tobacco might be one of the reasons for the increment of resistance in the transgenic tobacco.
Similar content being viewed by others
References
Wang, A. G., Oxygen metabolism in higher plants, in Plant Physiology and Molecular Biology (eds. Yu, S. W., Tang, C. Z.), Beijing: Science Press, 1998, 366.
Liang, Z., Ma, D. Q., Tang, L. et al., Expression of the spinach betaine aldehyde dehydrogenase (BADH) gene in transgenic to-bacco plants, Chinese J. Biotechnol., 1997, 13(3): 153.
Liu, F. H., Guo, Y., Gu, D. M. et al., Salt tolerance of transgenic plants with BADH cDNA, Acta Genetica Sinica, 1997, 24(1): 54.
Hayashi, H., Murata, N., Genetically engineering enhancement of salt tolerance in higher plants, in Stress Responses of Photosynthesis Organisms (eds. Satoh, K., Murata, N.), New York: Elservier, 1998, 133.
Yi, Y. J., Liu, J. Y., Luo, A. L. et al., Changes of photosystem II and respiratory enzymes activity in transgenic tobacco enriched BADH gene, Acta Botanica Sinica, 1999, 41(9): 993.
Cakmak, I., Marschner, H., Magnesium deficiency and high light intensity enhance activities of superoxide dismutase, ascorbate peroxidase and glutathione reductase in bean leaves, Plant Physiol., 1992, 98(4): 742.
Liang, Z., Yu, C., Huang, A. H. C., Isolation of spinach leaf peroxisomes in 0.25 molar sucrose solution by Percoll density gradient centrifugation, Plant Physiol., 1982, 70(4): 1210.
Giannopolitis, C. N., Ries, S. K., Superoxide dismutase I, Occurrence in higher plants, Plant Physiol., 1997, 59(2): 309.
Jiao, D. M., Mass screening for rice germplasm tolerant to photoinhibition, Photosynthetica, 1992, 26(3): 399.
Hanson, A. D., May, A. M., Grumet, R. et al., Betaine synthese in Chenopodes: Localization in chloroplasts, Proc. Natl. Acad. Sci. USA, 1985, 82(11): 3678.
Nakamura, T., Yokota, S., Muramoto, Y. et al., Expression of abetaine aldehyde dehydrogenase gene in rice, a glycinebetaine nonaccumulator and possible localization of its protein in peroxisomes, Plant J., 1997, 11(5): 1115.
Luo, A. L., Zhao, Y., Li, Y. C. et al., Localization of betaine aldehyde dehydrogenase in the cell of spinach leaf, Acra Phytophysiologica Sinica, 1995, 21(2): 117.
Rathinasabapathi, B., McCue, K. F., Gage, D. A. et al., Metabolic engineering of glycine betaine synthese: Plant betaine aldehyde dehydrogenase lacking typical transit peptides are targeted to to-bacco chloroplasts where they confer betaine aldehyde resistance, Planta, 1994, 193(2): 155.
Hou, C. X., Xu, C. H., Tang, C. Z. et al., Selective protection of glycinebetaine on extrinsic polypeptides of PS II particles, Chinese Science Bulletin, 1997, 42: 1744.
Papageorgiou, G. C., Murata, N., The unusually strong stabilizing effects of glycine betaine on the structure and function of the oxygen-evolving photosystem II complex, Photosynthesis Research, 1995, 44(3): 143.
Liang, Z., Zhao, Y., Tang, L. et al., Protection effect of betaine on respiratory enzymes, Acta Botanica Sinica, 1994, 36(12): 947.
Tang, P. S., Regulation and control of multiple pathways of respiratory metabolism in relation to other physiological functions in higher plants, Acta Botanica Sinica, 1979, 21(2): 93.
Liang, Z., Liang, H. G., The respiratory metabolism in higher plants, in Plant Physiology and Molecular Biology (ed. Yu, S. W., Tang, Z. C.), Beijing: Science Press, 1998, 344.
Liu, J. Y., Liang, Z., Wang, X. C., Regulation and control of gene expression in plants, Chinese Bulletin of Botany, 1999, 16(1): 1.
Nuccio, M. L., Rhodes, D., McNeil, S. D. et al., Meyabolic enineering of plants for osmotic stress resistance, Current Opinion in Plant Biology, 1999, 2(2): 128.
Smironoff, N., Plant resistance to enviromental stress, Current Opinion in Biotechnology, 1998, 9(2): 214.
Author information
Authors and Affiliations
About this article
Cite this article
Luo, A., Liu, J., Ma, D. et al. Increment of antioxidase activity of transgenic tobacco with betaine aldehyde dehydrogenase. Chin.Sci.Bull. 46, 492–495 (2001). https://doi.org/10.1007/BF03187265
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03187265