Abstract
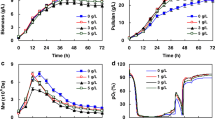
Effects of different pH and carbon sources on pullulan production, UDP-glucose level and pullulan-related synthases activity inAureobasidium pullulans Y68 were examined. It was found that more pullulan was produced when the yeast strain was grown in the medium with initial pH 7.0 than when it was grown in the same medium with constant pH 6.0. The results also show that higher pullulan yield was obtained when the cells were grown in the medium containing glucose than when they were cultivated in the medium supplementing other carbon sources. Our results demonstrate that the more pullulan was synthesized, the less UDP-glucose was left in the cells ofA. pullulans Y68. However, it was observed that more pullulan was synthesized; the cells had higher pullulan-related synthase activity. Therefore, high pullulan yield was related to low UDP-glucose level and high pullulan-related synthases activity inAureobasidium pullulans Y68.
Similar content being viewed by others
References
Alban S., Schauerte A., Franz G. (2002). Anticoagulant sulfated polysaccharides: part I: Synthesis and structure-activity relationships of new pullulan sulfates. Carbohydr. Polym., 47: 267–276.
Auer D.P.F., Seviour R.J. (1990). Influence of varying nitrogen sources on polysaccharide production byAureobasidium pullulans in batch culture. Appl. Microbiol. Biotechnol., 32: 637–644.
Bradford M.M. (1976). A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the princible of protein-dye binding. Anal. Biochem., 72: 248–253.
Catley B.J., McDowell W. (1982). Lipid-linked saccharides formed during pullulan biosynthesis inAureobasidium pullulans. Carbohydr. Res., 103: 65–75.
Chi Z.M., Liu J., Zhang W. (2001). Trehalose accumulation from soluble starch bySaccharomycopsis fibuligera sdu. Enzyme Microb. Technol., 28: 240–245.
Chi Z.M., Zhao S.Z. (2003). Optimization of medium and cultivation conditions for pullulan production by a new pullulan-producing yeast. Enzyme Microb. Technol., 33: 206–211.
Chi Z., Ma C., Wang P., Li H.F. (2007). Optimization of medium and cultivation conditions for alkaline protease production by the marine yeastAureobasidium pullulans. Biores. Technol., 98: 534–538.
Daran J.M., Dalles N., Thines-Sempoux D., Paquet V., Francois J. (1995). Genetic and biochemical characterization of the UGP1 gene encoding the UDP-glucose pyrophosphorylase fromSaccharomyces cerevisiae. Eur. J. Biochem., 233: 520–530.
Degeest B., Vuyst L.D. (2000). Correlation of activities of the enzymes α-phosphoglucomutase, UDP-galactose 4-epimerase and UDP-pyrophosphorylase with exopolysaccharide biosynthesis byStreptococcus thermophilus LY03. Appl. Environ. Microbiol., 66: 3519–3527.
Deshpande M.S., Rale V.B., Lynch J.M. (1992).Aureobasidium pullulans in applied microbiology: a status report. Enzyme Microb. Technol., 14: 514–527.
Grobben G.J., Smith M.R., Sikkema J.J., de Bont A.M. (1996). Influence of fructose and glucose on the production of exopolysaccharides and the activities of enzymes involved in the sugar metabolism and the synthesis of sugar nucleotides inLactobacillus delbrueckii subsp.bulgaricus NCFB 2772. Appl. Microbiol. Biotechnol., 46: 279–284.
Kurtzman C.P., Fell J.W., Eds (1998). The Yeasts, a taxonomic study, 4th edn Elsevier Science B.V., Amsterdam.
Lazaridon A., Roukas T., Biliaderis C.G., Varikousi H. (2002). Characterization of pullulan produced from beet molasses byAureobasidium pullulans in a stirred tank reactor under varying agitation. Enzyme Microb. Technol., 31: 122–132.
Lee J.H., Kim J.H.K., Zhu Z.I., Zhan X.B., Lee J.W., Shin D.H., Kim S.K. (2001). Optimization of conditions for the production of pullulan and high molecular weight pullulan byAureobasidium pullulans. Biotechnol. Lett., 23: 817–820.
Ma X., Joachim S. (2001). High yielding one-pot enzyme-catalyzed synthesis of UDP-glucose in gram scales. Carbohydr. Res. 333: 159–163.
Roukas T., Liakopoulou-Kyriakides M. (1999). Production of pullulan from beet molasses byAureobasidium pullulans in a stirred tank fermentor. J. Food Eng., 40: 89–94.
Shingel K.I. (2004). Current knowledge on biosynthesis, biological activity, and chemical modification of the exopolysaccharide, pullulan. Carbohy. Res., 339: 447–460.
Spiro R.G. (1966). Analysis of sugars found in glycoproteins. Method Enzymol., 8: 3–26.
Strominger J.L., Maxweil E.S., Kalckar H.M. (1957). Determination of UDPG and UTP by means of UDPG dehydrogenase. Method Enzymol., 3: 974–977.
Sutherland L.W. (1998). Novel and established application of microbial polysaccharide. Trends Biotechnol., 16: 41–46.
Taguchi R., Sakano Y., Kikuchi Y., Sakuma M., Kobayashi T. (1973). Synthesis of pullulan by acetone-dried cells and cell-free enzyme fromPullularia pullulans, and the participation of lipid intermediate. Agr. Biol. Chem., 37: 1635–1641.
Vijayendra S.V.N., Bansal D., Prasad M.S., Nand K. (2001). Jaggery: a novel substrate for pullulan-production byAureobasdium pullulans CFR-77. Proc. Biochem., 37: 359–364.
Yuen S. (1974). Pullulan and its applications. Proc. Biochem., 22: 7–9.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Duan, X., Chi, Z., Li, H. et al. High pullulan yield is related to low UDP-glucose level and high pullulan-related synthases activity inAureobasidium pullulans Y68. Ann. Microbiol. 57, 243–248 (2007). https://doi.org/10.1007/BF03175214
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03175214