Summary
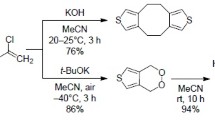
8-Chloro-1-thionaphthol, prepared fromperi-acid, was condensed with bromoacetaldehyde dimethyl acetal to give 8-chloro-1-naphthylω-dimethoxyethyl sulphide (IV). Ring-closure of (IV) gave 3′-chlorobenzo-(1′:2′, 6:7)-thionaphthene (V), which on dechlorination gave 6:7-benzothionaphthene (II). The cyclization product fromα-naphthylω-dimethoxyethyl sulphide (I) reported in the previous communication being different from (II) is therefore naphtho-(1′:8′-bc)-thiapyran (III).
9-Chloronaphtho-(1′:8′-bc)-thiapyran (VII) was prepared from Tobias acid in a manner similar to (V), but dechlorination of (VII) to (III) could not be carried out due to the low yields of (VII) in the cyclization of 2-chloro-1-naphthylω-dimethoxyethyl sulphide (VI).
The literature on the cyclization of certain relatedα-substituted naphthyl, naphthoxy and thionaphthoxy derivatives is discussed.
Similar content being viewed by others
References
Szmuzkovicz and Modest..J. Am. Chem. Soc., 1950,72, 571.
..DRP., 230, 237.
Johnson..Organic Reactions, 1944, Vol.II, p. 125 (John Wiley).
Inghamet al. ..J. Chem. Soc., 1931, 895.
Ullmann..Ber., 1897,30, 1468.
Anand and VenkataramanProc. Ind. Acad. Sci., 1948,28A, 160.
Friedländer and WoroschzowAnnalen, 1912,388, 18.
Harley-Mason and MannJ. Chem. Soc., 1942, 404.
Stoermer..Annalen, 1900,312, 310.
Tilak..Proc. Ind. Acad. Sci., 1950,32A, 390.
Dosser and Richter..J. Am. Chem. Soc., 1934,56, 1132.
Harley-Mason and MannJ. Chem. Soc., 1942, 404.
Author information
Authors and Affiliations
Additional information
Communicated by Dr. K. Venkataraman,f.a.sc.
Rights and permissions
About this article
Cite this article
Dikshit, V.K., Tilak, B.D. A new synthesis of thiophenes and thiapyrans. Proc. Indian Acad. Sci. (Math. Sci.) 33, 78 (1951). https://doi.org/10.1007/BF03172186
Received:
DOI: https://doi.org/10.1007/BF03172186