Abstract
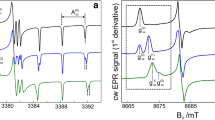
The structure and conformation of carboxylic acid, formyl, and propenoic acid derivatives of the nitroxyl spin-label 2,2,5,5-tetramethyl-1-oxypyrroline have been determined by electron nuclear double resonance (ENDOR) spectroscopy. From ENDOR spectra of the spin-label compounds in frozen solutions, we have assigned the resonance absorption features for each class of protons. The ENDOR spectra were analyzed on the basis of their dependence onH 0. The maximum and minimum ENDOR shifts for each proton were shown to correspond to axially symmetric principal hyperfine coupling (hfc) components, from which the dipolar contributions were estimated to calculate electron-proton separations. Conformational analysis on the basis of torsion angle search calculations constrained by the ENDOR determined electron-proton distances revealed that in all three spin-label compounds the side chains are in a planar conformation with respect to the oxypyrrolinyl ring. In the carboxylic acid and formyl derivatives the C=O group is in as-trans conformation with respect to the vinyl group of the spin-label, while in the spin-labeled propenoic acid the conformation is found to be all planartrans-s-cis.
Similar content being viewed by others
References
Hamilton C.L., McConnell H.M. in: Structural Chemistry and Molecular Biology (Rich A., Davidson N., eds.), pp. 115–149. San Francisco: W.H. Freeman 1968.
McConnell H.M., Gaffney-McFarland B.: Q. Rev. Biophys.3, 91 (1970)
Berliner L.J. (ed.) in: Spin Labeling: Theory and Application. New York: Academic Press 1976.
Hayat H., Silver B.L.: J. Phys. Chem.77, 72 (1973)
Davis T.D., Christofferson R.E., Maggiora G.M.: J. Am. Chem. Soc.97, 1347 (1975)
Wells G.B., Makinen M.W.: J. Am. Chem. Soc.110, 6343 (1988)
Mustafi D., Sachleben J.R., Wells G.B., Makinen M.W.: J. Am. Chem. Soc.112, 2558 (1990)
Wells G.B., Mustafi D., Makinen M.W.: J. Am. Chem. Soc.112, 2566 (1990)
Mustafi D., Boisvert W.E., Makinen M.W.: Biopolymers29, 45 (1990)
Mustafi D., Wells G.B., Joela H., Makinen M.W.: Free Radical Res. Commun.10, 95 (1990)
Joela H., Mustafi D., Fair C.C., Makinen M.W.: J. Phys. Chem. (in press).
Rozantsev E.G.: Free Nitroxyl Radicals, Chap. 9, pp. 205–206. New York: Plenum Press 1970.
Koch T.R., Kuo L.C., Douglas E.G., Jaffer S., Makinen M.W.: J. Biol. Chem.254, 12310 (1979)
Yim M.B., Makinen M.W.: J. Magn. Reson.70, 89 (1986)
Mustafi D., Makinen M.W.: Inorg. Chem.27, 3360 (1988)
Turley J.W., Boer F.P.: Acta Crystallogr. Sect. B28, 1641 (1972)
Boeyens J.C.A., Kruger G.J.: Acta Crystallogr. Sect. B26, 668 (1970)
Becher J., Svendsen E.N., Simonsen O.: Tetrahedron33, 1481 (1977)
Raghunathan S., Pattabhi V.: Acta Crystallogr. Sect. B37, 1299 (1981)
Coppens P.: Science158, 1577 (1967)
Jones T.A. in: Computational Crystallography (Sayre D., ed.), pp. 303–317. Oxford: Clarendon Press 1982.
Jones T.A.: Methods Enzymol.115, 157 (1985)
Marshall G.R.: personal communication. Detailed information on the use of this program package can be obtained from Tripos Associates, Inc., 1600 S. Hanley Road, St. Louis, Missouri 63144.
Naruto S., Motoc J., Marshall G.R., Daniels S.B., Sofia M.J., Katzenellenbogen J.A.: J. Am. Chem. Soc.107, 5262 (1985)
Iijima H., Dunbar J.B., Marshall G.R.: Proteins: Struct., Funct., Genet.2, 330 (1987)
Mustafi D., Joela H., Makinen M.W.: J. Magn. Reson.91, 497 (1991)
Wiberg K.B., Laidig K.E.: J. Am. Chem. Soc.109, 5935 (1987)
Forbes W.F., Shilton R.: J. Am. Chem. Soc.81, 786 (1959)
Wagner R., Fine J., Simmons J.W., Goldstein J.H.: J. Chem. Phys.26, 634 (1957)
Kashino S., Haisa M.: Acta Crystallogr. Sect. B36, 346 (1980)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mustafi, D., Makinen, M.W. Structure and conformation of nitroxyl spin-label compounds in frozen solutions by electron nuclear double resonance spectroscopy. Appl. Magn. Reson. 3, 321–331 (1992). https://doi.org/10.1007/BF03166701
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03166701