Abstract
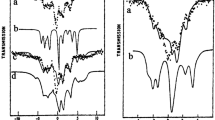
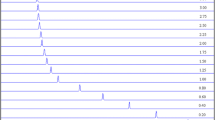
W-band (95 GHz) pulsed electron nuclear double resonance (ENDOR) measurements were carried out to determine quantitatively the first coordination shell of Mn2+ with ADP and ATPγS. The intensity of the ENDOR effect was used for counting the number of equivalent phosphate oxygens and water ligands. Titration curves for determining the binding constant of Mn2+. ADP were obtained using the intensity of the X-band EPR spectrum and the31P ENDOR effect. Both curves gave the same binding constant showing that phosphate ligand counting is plausible, provided that an appropriate reference is available. The comparison of the31P ENDOR effect of the 1:1 ADP and ATPγS complexes shows that two phosphates are coordinated in both; while in ADP they are equivalent, in ATPγS they are slightly different. The reference system for water ligand counting was Mn(H2O) 2+6 in a H2O-D2O mixture. The results show a smaller error for the2H ENDOR effect, compared to the1H ENDOR effect. Unlike the31P ENDOR effect, the1H ENDOR effect dependence on [ADP] in the titration experiments showed that it is sensitive to variations in the zero-field splitting, which in turn alters the contributions of transitions other than the ‖−1/2>↔‖1/2>. This results in a larger error in the determination of the number of water ligands.
Similar content being viewed by others
References
Reed G.H., Poyner R.R. in: Manganese and Its Role in Biological Processes. Metal Ions in Biological Systems, vol. 37 (Sigel A., Sigel H., eds.), pp. 183–207. New York: Marcel Dekker 2000.
Petersen J., Gessner C., Fisher A., Lowe D.J., Lubitz W.: Biochem. J.391, 527–539 (2005)
Tan X., Poyner R., Scholes C.P., Reed G.H.: Biochemistry32, 7799–7810 (1993)
Zoleo A., Contessi S., Lippe G., Pinato L., Brustolon M., Brunel L.C., Dabbeni-Sala F., Maniero A.L.: Biochemistry43, 13214–13224 (2004)
Buy C., Girault G., Zimmermann J.-L.: Biochemistry35, 9880–9891 (1996)
Zimmermann J.-L., Schneider B., Morlet S., Amano T., Sigalat C.: Spectrochim. Acta A56, 285–299 (2000)
Bennati M., Hertel M.M., Fritscher J., Prisner T.F., Weiden N., Hofweber R., Sporner M., Horn G., Kalbitzer H.R.: Biochemistry45, 42–50 (2006)
Legler P.M., Lee H.C., Peisach J., Mildvan A.S.: Biochemistry41, 4655–4668 (2002)
Morrissey S.R., Horton T.E., DeRose V.J.: J. Am. Chem. Soc.122, 3473–3481 (2000)
Hoogstraten C.G., Grant C.V., Horton T.E., DeRose V.J., Britt R.D.: J. Am. Chem. Soc.124, 834–842 (2002).
Halkides C.J., Farrar C.T., Larsen R.G., Redfield A.G., Singel D.J.: Biochemistry33, 4019–4035 (1994)
Rohrer M., Prisner, T. F., Bruegmann O., Kaess H., Spoerner M., Wittinghofer A., Kalbitzer H.R.: Biochemistry40, 1884–1889 (2001)
Larsen R.G., Halkides C.J., Redfield A.G., Singel D.J.: J. Am. Chem. Soc.114, 9608–9611 (1992)
Schneider B., Sigalat C., Amano T., Zimmermann J.-L.: Biochemistry39, 15500–15512 (2000)
Eccleston J.F., Webb M.R., Ash D.E., Reed G.H.: J. Biol. Chem.256, 10774–10777 (1981)
Leyh T.S., Sammons R.D., Frey P.A., Reed G.H.: J. Biol. Chem.257, 15047–15053 (1982)
Olsen L.R., Reed G.H.: Arch. Biochem. Biophys.304, 242–247 (1993)
Reed G.H., Leyh T.S.: Biochemistry19, 5472–5480 (1980)
Astashkin A.V., Raitsimring A.M.: J. Chem. Phys.117, 6121–6132, (2002)
Hoogstraten C.G., Britt R.D.: RNA8, 252–260 (2002)
Zech S.G., Sun W.-C., Jacques V., Caravan P., Astashkin A.V., Raitsimring A.M.: Chem. Phys Chem.6, 2570–2577 (2005)
Raitsimring A.M., Astashkin A.V., Baute D., Goldfarb D., Caravan P.: J. Phys. Chem. A108, 7318–7323 (2004)
Vardi R., Bernardo M., Thomman H., Strohmaier K.G., Vaughan D.E.W., Goldfarb D.: J. Magn. Reson.126, 229–241 (1997)
Astashkin A.V., Raitsimring A.M., Caravan P.: J. Phys. Chem. A108, 1990–2001 (2004)
Arieli D., Delabie A., Groothaert M., Pierloot K., Goldfarb D.: J. Phys. Chem. B106, 9086–9097 (2002)
Gromov I., Krymov V., Manikandan P., Arieli D., Goldfarb D.: J. Magn. Reson.139, 8–17 (1999).
Epel B., Arieli D., Baute D., Goldfarb D.: J. Magn. Reson.164, 78–83 (2003)
Tan X.L., Bernardo M., Thomann H., Scholes C.P.: J. Chem. Phys.98, 5147–5157 (1993)
Epel B.; Manikandan P., Kroneck P.M.H., Goldfarb D.: Appl. Magn. Reson.21, 287–297 (2001)
Jallon J., Cohn M.: Biochim. Biophys. Acta222, 542–545 (1970)
Eckstein F.: Annu. Rev. Biochem.54, 367–402 (1985)
Zetter M.S., Lo G.Y.S., Dodgen H.W., Hunt J.P.: J. Am. Chem. Soc.100, 4430–4436 (1978)
Mustafi D., Telser J., Makinen M.W.: J. Am. Chem. Soc.114, 6219–6226 (1992)
Sternlicht H., Shulman R.G., Anderson E.W.: J. Chem. Phys.43, 3123–3132 (1965)
Baute D., Goldfarb D.: J. Phys. Chem. A109, 7865–7871 (2005)
Serpersu E.H., McCracken J., Peisach J., Mildvan A.S.: Biochemistry27, 8034–8044 (1988)
Walsby C.J., Telser J., Rigsby R.E., Armstrong R.N., Hoffman B.M.: J. Am. Chem. Soc.127, 8310–8319 (2005)
LoBrutto R., Smithen G.W., Reed G.H., Orme-Johnson W.H., Tan S.L., Leigh J.S.: Biochemistry25, 5654–5660 (1986)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Potapov, A., Goldfarb, D. Quantitative characterization of the Mn2+ complexes of ADP and ATPγS by W-band ENDOR. Appl. Magn. Reson. 30, 461–472 (2006). https://doi.org/10.1007/BF03166212
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF03166212