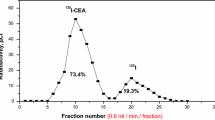
Abstract
A total of 7 (4 males and 3 females) patients were included in this retrospective study to determine the sensitivity of radioimmunoscintigraphy with I-131 labeled anti CEA/CA 19-9 monoclonal antibodies. Out of 7 patients 2 had ascending colon cancer, one had sigmoid colon cancer, one had rectal cancer and one had adenocarcinoma in the CBD and the remaining two had metastatic tumor (one in the lungs and the other in the liver). Whole body as well as spot images showed a 72% (5/ 7) positive scan. But post operative specimen counts and imaging showed a high tumor to nontumor ratio and a good tumor to non-tumor contrast of activity of I-131 labeled monoclonal antibody. We did not find any relation between CEA/CA 19-9 levels and scan findings. A case of liver metastasis was also detected by this radioimmnoscintigraphy.
Similar content being viewed by others
References
Larson SM. Clinical radioimmunodetection. 1978–1988: over view and suggestions for standardization of clinical trials.Cancer Res 50 (suppl): 892–898, 1990.
Gold P, Freedman SO. Demonstration of tumor specific antigen in human colonic carcinoma by immunological tolerance and absorption technique.J Exp Med 121: 439–459, 1965.
Shively JE, Betty JD. CEA related antigens: molecular biology and clinical significance.Crit Rev Anc Hematol 2: 355–399, 1985.
Jasson BA, Strand SE, Andersson L. Radiation dosimetry for In-111 labeled anti CEA-F(ab′)2 fragments evaluated from tissue distribution in rats.J Nucl Med 33: 1654—1660, 1992.
Goldenberg DM, Goldenberg H, Sharkey RM, Lee RE, Ford EH, Horowitz JA, et al. Imaging of colorectal carcinoma with radio labeled antibodies.Semin Nucl Med 19: 262–281, 1989.
Hinkle GH, Loesch JA, Hill TL, Lefevre SR, Olson JO. Indium-111 monoclonal antibodies in radioimmunoscintigraphy.J Nucl Med Technol 18: 16–28, 1990.
Baum RP, Maul FD, Senekowitsch R, Lorenz M, Hottenrott C, Happ J, et al. Radioimmunoscintigraphy of CA 19-9/ CEA producing tumours using I-131 labeled F(ab′)2 fragments of monoclonal antibodies (19-9/anti CEA radioimmuno cocktail).Nuclear Medicine in Clinical Oncology, C. Winkler (ed.), Springer-Verlag Berlin Heidelberg, pp. 197–206, 1986.
Goldenberg DM, Larson SM. Radioimmunodetection in cancer identification.J Nucl Med 33: 803–814, 1992.
Haseman MK, Brown DW, Keeling CA, Reed NL. Radioimmunodetection of occult carcinoembryonic antigen producing cancer.J Nucl Med 33: 1750–1757, 1992.
Carrasquillo JA, Sugarbaker P, Colcher D, Reynolds JC, Esteban J, Bryant G, et al. Radioimmunoscintigraphy of colon cancer with I-131 labeled B72.3 monoclonal antibody.J Nucl Med 29: 1022–1030, 1988.
Bhargava KK, Acharya SA: Labeling of monoclonal antibodies with radionuclides.Semin Nucl Med 19. 187–201, 1989.
Hnatowich JD, Griffin TW, Kosciuczyk C, Rusckowski M, Childs RL, Mattis JA, et al. Pharmacokinetics of an Indium-111 labeled monoclonal antibody in cancer patients.J Nucl Med 26: 849–858, 1985.
Hnatowich JD. Dissociation and radiolabeling of antibodies.New Procedures in Nuclear Medicine, R.P. Spencer (ed.), CRC press, Florida, p. 145, 1989.
Perkins AC, Whalley DR, Ballantyne KC, Pimm MV. Gamma camera emission tomography using radiolabelled antibodies.Eur J NuclMed 14: 45–49, 1988.
Zimmer AM, Rosen ST, Spics SM, Leikin RG, Kazikiewicz JM, Silverstein EA, et al. Radioimmunotherapy of patients with cutaneous T-celllymphoma using an Iodine-131 labeled monoclonal antibody: analysis of retreatment following plasmapheresis.J NuclMed 29: 174–180, 1988.
Halpern SE, Tarburton JP, Sudora E, Hagan P. Alterations in an indium-111 Fab′ under conditions of utilization.Eur J NuclMed 19: 387–393, 1992.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Arbab, A.S., Koizumi, K., Uchiyama, G. et al. Radioimmunoscintigraphy of CEA/CA 19-9 producing tumors with I-131 labeled monoclonal antibodies. Ann Nucl Med 8, 9–15 (1994). https://doi.org/10.1007/BF03164981
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03164981