Abstract
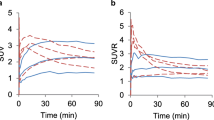
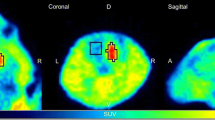
Using positron emission tomography and radio-high performance liquid chromatography, the accumulation of 2′-deoxy-5-18F-fluorouridine in the brain tumors and plasma pharmacokinetic parameters were investigated in 20 patients. High accumulation of the tracer in high grade gliomas and meningiomas and very rapid degradation of the tracer in the plasma were found. Very large variations were observed in both tumor accumulation and pharmacokinetic data. The tumor accumulation, however, did not correlate with any of the plasma pharmacokinetic parameters: area under the plasma concentration-time curve, mean residence time, total body clearance and steady-state volume of distribution. The results suggest that the accumulation of the tracer reflects the metabolic activity of the brain tumor tissues and that the effect of the rapid metabolic change in the tracer in the plasma on the tumor accumulation may be minor.
Similar content being viewed by others
References
Heiderberger C, Danengerg PV, Moran R: Fluorinated pyrimidin.es and their nucleoside.Adv Enzymol 54: 57–119, 1983
Abe Y, Fukuda H, Ishiwata K, et al: Studies on18F-labeled pyrimidines. Tumor uptake of18F-5-fluorouracil,18F-5-fluorouridines and18F-5-fluorodeoxy-uridine in animals.Eur J Nucl Med 8: 258–261, 1983
Ishiwata K, Ido T, Kawashima K, et al: Studies on18F-labeled pyrimidines IL Metabolic investigation of18F-5-fluorouracil,18F-5-fluoro-2′-deoxyuridine and18F-5-fluorouridine in animais.Eur J Nucl Med 9: 185–189, 1984
Ishiwata K, Ido T, Abe Y, et al: Studies on18F-labeled pyrimidines III. Biological investigation of18F-labeled pyrimidines and comparison with3H-deoxythimidine in tumor-bearing rats and mice.Eur J Nucl Med 10: 39–44, 1985
Abe Y, Matsuzawa T, Itoh M, et al: Cancer detection with18F-5-fluorodeoxyuridine—a new cancer diagnosis agent reflecting the nucleic acid metabolism.Jpn J Nucl Med 22: 583–586, 1985 (in Japanese)
Kiyosawa M, Mizumo K, Ishiwata K, et al: Orbital tumor diagnosis by positron emission tomography using18F-fiuorodeoxyuridine.Ophthalmic Res 18: 292–298, 1986
Kameyama M, Tsurumi Y, Itoh J, et al: Biological malignancy and 18FdUrd uptake in glioma patients —PET study of nucleic acid metabolism.CYRIC Annual Report, Tohoku University, 1988: 215–227, 1988
Kameyama M, Tsurumi Y, Shirane R, et al: Nucleic acid metabolism in glioma studied with18FdUrd and PET. JCereb Blood Flow Metab 9 (suppl 1): S221, 1989
Tsurumi Y, Kameyama M, Ishiwata K, et al:18F-fluoro-2′-deoxyuridine as a tracer of nucleic acid metabolism in brain tumors.J Neurosurg 72: 110–113, 1990
Ishiwata K, Sato K, Kameyama M, et al: Metabolic fates of 2′-deoxy-5-[18F]fiuorouridme in tumor-bearing mice and human plasma.Nucl Med Bio 118: 539–545, 1991
Kubota K, Ishiwata K, Kubota R, et al: Tracer feasibility for monitoring tumor radiotherapy: A quadruple tracer study with fiuorine-18-fluorodeoxyglucose or fluorine-18-fluorodeoxyuridine, L-[methyl-14C]methionine, [6-3H]thymidine, and gallium-67.J Nucl Med 32: 2118–2123, 1991
Ishiwata K, Takahashi T, Iwata R, et al: Tumor diagnosis by PET: Potential of seven tracers examined in five experimental tumors including an artificial metastasis model.Nucl Med Biol 19: 611–618, 1992
Sato K, Kameyama M, Ishiwata K, et al: Metabolic changes of glioma following chemotherapy: An experimental study using PET tracers.J Neuro-Oncol 14: 81–89, 1992
Ishiwata K, Monma M, Iwata R, et al: Automated synthesis of 5-[18F]fluoro-2′-deoxyuridine.Appl Radiat Isot 38: 467–473, 1987
Yamaoka K, Tanigawara Y, Nakagawa T, et al: Capacity-limited elimination of cefmetazole in rat.Int J Pharmaceu 10: 291–300, 1978
Parlak CS, Blasberg RG, Fenstermacher JD: Graphical evaluation of blood-to-brain transfer constants from multi-time uptake data.J Cereb Blood Flow Metab 3: 1–7, 1983
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ishiwata, K., Tsurumi, Y., Kameyama, M. et al. Brain tumor accumulation and plasma pharmacokinetic parameters of 2′-deoxy-5-18F-fluorouridine. Ann Nucl Med 7, 199–205 (1993). https://doi.org/10.1007/BF03164967
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03164967