Abstract
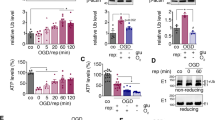
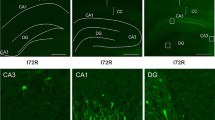
Ubiquitin is involved in the degradation of denatured proteins in the recovery process after various stresses. To clarify the different responses of the ubiquitin system in the hippocampal neurons after ischemia, we chose 7.5 min of sublethal forebrain ischemia in the rat. After 7.5 min of ischemia, ubiquitin-like immunoreactivity (UIR) in most of the hippocampal pyramidal cells, except for the interneurons, diminished after 3 h of reperfusion, but enhanced UIR and subsequent recovery of UIR were observed in the different hippocampal regions after 24 h of reperfusion. The most prolonged recovery of UIR in the hippocampal cells was observed in the CA1 neurons after 72 h of reperfusion. Immunoblot analysis of the proteins extracted from CA1 region showed that high-mol-wt ubiquitin conjugates (HMWUC) above 40 kDa increased, whereas free ubiquitin and ubiquitinated histone 2A decreased slightly after 4 h and 24 h of reperfusion. At 72 h of reperfusion, HMWUC decreased to the original level and free ubiquitin slightly increased beyond the control level. These results suggested that (1) diminished UIR does not always mean depletion of entire ubiquitin-protein conjugates; (2) even after sublethal ischemia, damaged proteins in the CA1 neurons may increase, and it may take a long time for elimination of these proteins.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bensadoun A., and Weinstein D. (1976) Assay of proteins in the presence of interfering materials.Anal. Bochem. 70, 241–250.
Finley D. and Varshavsky A. (1985) The ubiquitin system: Functions and mechanisms.Trend. Biochem. Sci. 10, 343–347.
Ishibashi Y., Takada K., Joh K., Ohkawa K., Aoki, T., and Matsuda M. (1991) Ubiquitin immunoreactivity in human malignant tumours.Br. J. Cancer 63, 320–322.
Kirino T. (1982) Delayed neuronal death in the gerbil hippocampus following ischemia.Brain Res. 239, 57–69.
Kitagawa K., Matsumoto M., Tagaya M., Hata R., Ueda H., Niinobe M., Handa N., Fukunaga R., Kimura K., Mikoshiba K., and Kamada, T. (1990) “Ischemic tolerance” phenomenon found in the brain.Brain Res.528, 21–24.
Laemmli U. K. (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4.Nature 227, 680–685.
Leigh P. N., Probst A., Dale G. E., Power D. P., Brion J.-P., Dodson A., and Anderton B. H. (1989) New aspects of the pathology of neurodegenerative disorders as revealed by ubiquitin antibodies.Acta Neuropathol. (Berlin)79, 61–72.
Lowry O. H., Rosebrough N. J., Farr A. L., and Randall R. J. (1951) Protein measurement with the Folin phenol reagent.J. Biol. Chem. 193, 265–275.
Magnusson K. and Wieloch T. (1989) Impairment of protein ubiquitination may cause delayed neuronal death.Neurosci. Lett. 96, 264–270.
Manetto V., Perry G., Tabaton M., Mulvihill P., Fried V. A., Smith H. T., Gambetti P., and Autilio-Gambetti L. (1988) Ubiquitin is associated with abnormal cytoplasmic filaments characteristic of neurodegenerative diseases.Proc. Natl. Acad. Sci. USA 85, 4501–4505.
Pulsinelli W. A. and Brierley J. B. (1979) Anew model of bilateral hemispheric ischemia in the unanesthetized rat.Stroke 10, 267–272.
Pulsinelli W. A., Brierley J. B., and Plum F. (1982) Temporal profile of neuronal damage in a model of transient forebrain ischemia.Ann. Neurol. 11, 491–498.
Pulsinelli W. A. and Duffy T. E. (1983) Regional energy balance in rat brain after transient forebrain ischemia.J. Neurochem. 40, 1500–1503.
Swerdlow P. S., Finley D., and Varshavsky A. (1986) Enhancement of immunoblot sensitivity by heating of hydrated filters.Anal. Biochem. 156, 147–153.
Vass K., Welch W. J., and Nowak T. S., Jr. (1988) Localization of 70-kDa stress protein induction in gerbil brain after ischemia.Acta Neuropathol. (Berlin)77, 128–135.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hayashi, T., Takada, K. & Matsuda, M. Changes in ubiquitin and ubiquitin-protein conjugates in the CA1 neurons after transient sublethal ischemia. Molecular and Chemical Neuropathology 15, 75–82 (1991). https://doi.org/10.1007/BF03161057
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03161057