Abstract
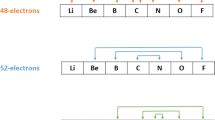
An elementary method (the so-called “chain method”) [1] is applied to the evaluation of the Madelung constant of crystal lattices both of the NaCl and the CsCl-type. The binding energies are summed up over suitably chosen cylinder shells resulting in very good approximate values for the Madelung constant. The Madelung constants both of CaF2 and ZnS-type lattices are determined from linear relations given byBenson andvan Zeggeren [2].
Резюме
Соответствующим применением цепного метода автором вычисляется постоянная Маделунга для решёток NaCl и CsCl. На основе этих результатов, используя линейные зависимости Бензона и Цеггерена, определяется значение постоянной Маделунга для решёток CaF2 и ZnS.
Similar content being viewed by others
References
F. Fáthy andF. Bukovszky, Acta Phys. Hung.,8, 89, 1957;9, 275, 1959.
G. C. Benson andF. van Zeggeren, J. Chem. Phys.,26, 1083, 1957.
E. Madelung, Phys. Zs.,19, 524, 1918.
W. Kossel, Die molekularen Vorgänge beim Kristallwachstum, Leipziger Vorträge, Hirzel, Leipzig, 1928.H. E. Buckley, Crystal Growth, John Wiley and Sons, New York, 1951.
F. C. Frank, Phil. Mag.,41, 1287, 1950.
O. Emersleben, Der Wert der Madelungkonstanten des Steinsalzgitters. Kritische Zusammenfassung. Wiss. Zs. d. Univ. Greifswald, III, 607, 1953/54.Y. Sakamoto, J. Sci. Hiroshima Univ. Ser. A.,16, 569, 1953.
R. Hoppe, Z. Anorg. Allgem. Chem.,283, 196, 1956.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bukovszky, F. Elementary calculations of the Madelung constants of some cubic lattices. Acta Physica 18, 157–172 (1965). https://doi.org/10.1007/BF03156696
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03156696