Abstract
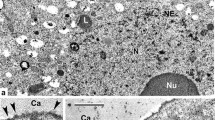
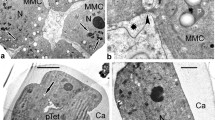
The embryo sac ofNigella damascena, with wall ingrowths over entire surface and distinctly formed wandlabyrinthe at the poles, displays a well integrated model for short distance transport of nutrients. The degenerating nucellus creates an influx of metabolites into the embryo sac. The embryo sac wandlabyrinthe play a dual role—initially aid with the influx of metabolites into the embryo sac, but later participate in endosperm wall formation. The two synergid apices exhibit prominent filiform apparatus which function differently. One of the synergids degenerates immediately after fertilization where filiform apparatus helps in receiving the pollen tube. The other undamaged synergid persists along with filiform apparatus upto the octant embryo stage and this filiform apparatus probably facilitates short distance metabolite transport into the synergid. The antipodal cells show 3 types of wall ingrowths—(i) wall ingrowths at the antipodal-nucellus interface which are long, branched and spread fan-like into the antipodal cytoplasm, indicating the flow of metabolites into the antipodes from the nucellus, (ii) inter-antipodal wall-ingrowths that are small, papillate and present on both sides of the wall indicating the exchange of metabolites between the antipodes and (iii) wall labyrinths at the antipode-central cell interance which are very small and are directed into the antipode cytoplasm.
Similar content being viewed by others
References
Bhandari N N and Sachdeva A 1985 Some aspects of organization and histochemistry of the embryo sac ofScilla sibirica Sato;Protoplasma 116 170–178
Bohdanowicz J and Turala-Szybowska K 1985 Ultrastructure of endopolyploid antipodals inAconitum vulparia Rchb. I. Antipodals in mature embryo sac;Protoplasma 127 163–170
Dumas A 1978 Le development de sac embryonnaire deConium maculatum L. et les phenomenes secretoires au moment de la fecondation;Bull. Soc. Bot. Fr. Act. Bot. 125 193–199
Feder N and O’Brien T P 1968 Plant microtechnique: Some principles and new methods;Am. J. Bot. 55 123–142
Fisher D B 1968 Protein staining of ribboned epon sections for light microscopy;Histochemie 16 92–96
Gunning B E S and Pate J S 1969 “Transfer cells”—Plant cells with wall ingrowths, specialised in relation to short distance transport of solutes—their occurrence, structure and development;Protoplasma 68 107–133
Gunning B E S and Pate J S 1974 Transfer cells; inDynamic aspects of plant ultrastructure (ed.) (A W Robards) (London: Mcgraw Hill) pp 441–480
Jensen W A 1962Botanical histochemistry (San Francisco: W A Freeman)
Maheshwari P 1950An introduction to the embryology of Angiosperms (New York: Mc-Graw Hill)
Mares D J, Stone B A, Jeffrey C and Norstog K 1977 Early stages in the development of wheat endosperm. II. Ultrastructural observations on cell wall formation;Am. J. Bot. 25 599–613
Marinos N G 1970 Embryogenesis of the Pea (Pisum sativum). 1. The cytological environment of the developing embryo;Protoplasma 70 261–279
Newcomb W 1973 The development of the embryo sac of sunflowerHelianthus annuus before fertilization;Can. J. Bot. 51 863–878
Newcomb W and Steeves T A 1971Helianthus annuus embryogenesis, embryo sac wall projections before and after fertilization;Bot. Gaz. 132 367–371
Newcomb W and Fowke L C 1973 The fine structure of the change from free nuclear to cellular condition in the endosperm of chickweedStellaria media;Bot. Gaz. 134 236–241
Prabhakar Kumkum and Vijayaraghavan M R 1983 Embryo sac wall inIberis amara—its ultrastructure and histochemistry;Phyton (Horn, Austria) 23 31–38
Rifot M 1971 L’evolution ultrastructurale des parois en rapport avec les transformations ontogeneques du gametophyte femelle chezAquilegia vulgaris L;Ann. Univ. A.R.E.R.S. 9 66–72
Rifot M 1973 Evolution structurale du Pole chalazien du sac. embryonnaire d’Aquilegia vulgaris en liasson avec son activite trophique;C.R. Acad. Sci. Paris 277 1313–1316
Schulz S R and Jensen W A 1968Capsella Embryogenesis: The synergids before and after fertilization;Am. J. Bot. 55 541–552
Schulz S P and Jensen W A 1969Capsella Embryogenesis: The suspensor and the basal cell;Protoplasma 67 139–163
Singh A P and Mogensen H L 1976 Fine structure of early endosperm inQuercus gambelii;Cytologia 41 345–361
Torosian C D 1971 Ultrastructural study of endosperm haustorial cell ofLobelia dunnii Greene (Campanulaceae, Lobelioideae);Am. J. Bot. 58 456–457
Vazart J 1969 Organization et ultrastructure du sac embryonnaire du Lin (Linum usitatissimum L.);Rev. Cytol. Biol. Veg. 32 227–240
Vijayaraghavan M R and Prabhakar Kumkum 1985 The Endosperm: inEmbryology of angiosperms (ed.) B M Johri (Berlin, Heidelberg, New York: Springer) pp 319–376
Vijayaraghavan M R and Bhat U 1984 Synergids before and after fertilization;Phytomorphology 33 74–84
Willemse M T H and Kapil R N 1981 Antipodes ofGasteria verrucosa (Liliaceae)—an ultrastructural study;Acta Bot. Neerl. 30 25–32
Yu S H and Chao C H 1979 Histochemical studies of ovary tissues during the embryo sac development inPaspalum longifolium Roxb;Caryologia 32 147–160
Zhukova G Y A and Sokolovskaya T B 1977 Ultrastructure of antipodes ofAconitum napellus (Ranunculaceae) embryo sac before fertilization;Bot. Zh. (Leningrad) 62 1600–1611
Author information
Authors and Affiliations
Additional information
Since deceased.
Rights and permissions
About this article
Cite this article
Vijayaraghavan, M.R., Misra, G. & Sujata, V. Wandlabyrinthe in the embryo sac ofNigella damascena Linn.. Proc. Indian Acad. Sci. 98, 261–268 (1988). https://doi.org/10.1007/BF03053797
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03053797