Summary
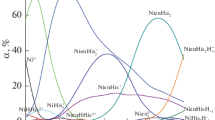
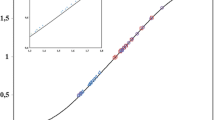
The composition of nickel ferrocyanide was investigated following the amperometric titration of Ni++ up to 40 mM. concentration against potassium ferrocyanide; it appeared to be 3 NiK2 [Fe (CN)6], Ni2 [Fe (CN)6] or Ni5K6 [Fe (CN)6]4. The data showed the possibility of estimating Ni++ polarographically within an experimental error of 1 per cent.
Similar content being viewed by others
References
WernerPharm. Zty., 1918,55, 211–12.
—Z. anal. Chem., 1919,58, 23–24.
Gaspar, Arnal and Alberto Castro, Girona Y. PozuramaAnales Soc. espan. fis. quim., 1926,24, 323–35.
Britton and Eric N. DoddJ. Chem. Soc., 1933, 1543–46.
Rene ParisCompt. rend., 1934,199, 863–65.
Harry, Weiser, Milligan and BatesJ. Phys. Chem., 1938,42, 945–54.
Sanigar, G.Trav. Chim. Pays. Bas., 1925,44, 59.
Ramaiah and AgarwalProc. Ind. Acad. Sci., 1956,44 A, 134.
—Ibid.,, 1956,44 A, 26.
NilssonChem. Zentr., 1942,1, 2972.
Kolthoff and Lingane Polarography, Interscience Publication, Inc., New York, 1952, 1.
Author information
Authors and Affiliations
Additional information
(Communicated by Prof.K. S. G. Doss, f.a.sc.)
Rights and permissions
About this article
Cite this article
Saraswat, H.C., Agarwal, S.K.D. Polarographic studies on the composition of nickel ferrocyanide complex. Proc. Indian Acad. Sci. 45, 91–96 (1957). https://doi.org/10.1007/BF03052785
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03052785