Abstract
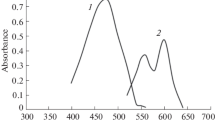
2′-Hydroxy chalcone gave an intense orange-yellow color with beryllium in the pH range 5·5–6·2. The limits of identification and dilution were 0·125γ and 1:4 × 105 respectively. A detailed spectro-photometric investigation of the color reaction was carried out. The complex obeyed Beer’s Law in the concentration range of 0·5–6·0p.p.m. at 460 nm. In the complex the ratio of metal to ligand is 1:2. The instability constant of the complex was 2·176 × 10−6 at 28°. Oxalate, tartrate, citrate and fluoride interfered. The tolerance limits of iron, aluminium, chromium, uranium, thorium and vanadium are reported.
Similar content being viewed by others
References
Job, P...Ann. Chim., 1928,9, 113;CA., 1928,22, 2120.
Edmonds, S. M. and Nathan BirnbaumJ. Am. Chem. Soc., 1941,63, 1471.
Lenseet al...Virginia J. of Science, 1942,3, 14;CA., 1942,36, 3113.
Tei-ichi Asahina..Bull. Chem. Soc., Japan, 1944,9, 133.
Harvey, A. E. and Manning, D. L.J. Am. Chem. Soc, 1950,72, 4488.
Snell, F. D., Snell, C. T. and Snell, C. A.Colorimetric Methods of Analysis, 1959,2A.
- and Snell, C. T.Ibid., 1961,2.
Vogel, A. I. ..A Text-Book of Qualitative Inorganic Analysis, Third Edition, 1962, p. 518.
I.U.P.A.C...Spectrophotometric Data for Colorimetric Analysis, Butterworths, London, 1963, pp. 37–43.
Author information
Authors and Affiliations
Additional information
Communicated by Prof. K. Neelakantam,f.a.sc.
Rights and permissions
About this article
Cite this article
Naidu, R.R. Spectrophotometric investigation of beryllium-2-hydroxy chalcone complex. Proc. Indian Acad. Sci. 75, 124–131 (1972). https://doi.org/10.1007/BF03049733
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03049733