Abstract
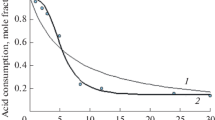
The alkaline hydrolysis of dicarboxylic esters have been studied in mixed solvent systems of dioxan-water and acetone-water. The two rate constants have been obtained by the Frost Schwemer method combined with the Laidler-Landskroner Transition state, the results are consistent with an ion dipole process for both steps. The results indicate that increasing distance between the carbethoxy groups leads to a limiting value for the ratio which for any given solvent composition range is not sensitive to specific influences.
Similar content being viewed by others
References
Anantakrishnan, S. V. and Rakhakrishnamurthi, P. S.Proc. Ind. Acad. Sci., 1962,56 A, 249.
Ingold, C. K.et al. ..J.C.S., 1930, 1032.
Anantakrishnan, S. V. and Venkatarathnam, R. V.Ind. J. Chem., 1966,4.
Evans, H. G. and Polanyi, M.Trans. Farad. Soc., 1936,32, 1333.
Goates, R. and Sullivan, R. J.J. Phys. Chem., 1958,62, 188.
Laidler, K. J. and Landskroner, P. A.Trans. Farad. Soc., 1956,52, 200.
Author information
Authors and Affiliations
Additional information
Based on the thesis of R.V.V. approved for the Ph.D. Degree of the Madras University, 1963.
Rights and permissions
About this article
Cite this article
Anantakrishnan, S.V., Venkatarathnam, R.V. Kinetic studies in ester hydrolysis. Proc. Indian Acad. Sci. 65, 188–194 (1967). https://doi.org/10.1007/BF03049530
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03049530