Summary
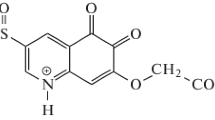
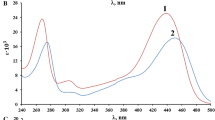
Absorption spectra of 4-(p-nitrophenyl-azo)-8-hydroxy-quinoline (NQOH) in the visible region was studied at different pH values in the range 4–12. NQOH exhibited two absorption maxima, one at λ = 480 mμ in acid solution and the other at λ = 600 mμ in alkaline solution. The absorption at λ = 600 mμ was markedly variant with the pH of the solution; the data indicated two dissociation processes presumably due to the ampholytic character of NQOH

with 7·3 and 8·1 for pK b and pK a referring in acidic and basic dissociation processes, respectively. The optical density values calculated from these constants agreed closely with the observed data.
Similar content being viewed by others
References
Gutzeit, G. and Monnier, R.Helv. Chim. Acta, 1933,16, 233, 478.
Niederl, B. J. and Niederl, V.Micromethods of Qualitative Organic Elementary Analysis, John Wiky, New York, 1938.
Boyd, J., Degering, E. F. and Shreve, R.N.Ind. Eng. Chem., 1938,10, 606.
Shreve, R. N. and Bennett, R.B.Jour. Amer. Chem. Soc., 1943,65, 2243.
Das, R. and Gutra Sirear, S.S.Jour. Ind. Chem. Soc., 1955,32, 679.
Vogel, A. I.Text-Book of Practical Organic Chemistry, Longmans, Green & Co., London.
Britton, H. T. S.Hydrogen Ions, Chapman and Hall, Ltd., London, 1932.
Author information
Authors and Affiliations
Additional information
Communicated by K. S. G. Doss, F.A.SC.
A preliminary note was published inNaturwiss., 1956,43, 179.
Rights and permissions
About this article
Cite this article
Ramaiah, N.A., Raman, V.S. Spectrophotometric determination of the dissociation constants of 4-(p-nitrophenyl-azo)-8-hydroxy-quinoline. Proc. Indian Acad. Sci. 44, 90–95 (1956). https://doi.org/10.1007/BF03048806
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03048806