Summary
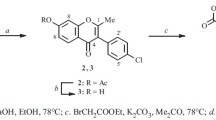
Using the more easily accessible 5:7: 8-trihydroxy derivatives of isoflavones a convenient method has been worked out for the synthesis of 5: 6: 7-trihydroxy derivatives. It consists in the alkali fission of 5-hydroxy-7: 8-dimethoxy isoflavone and its 2-methyl derivative to yield 2: 6-dihydroxy-3: 4-dimethoxy-phenyl-benzyl ketone. This ketone on treatment with ethyl formate and sodium yields 5-hydroxy-6 : 7-dimethoxy isoflavone. Vigorous acetylation of the ketone using acetic anhydride and sodium acetate yields 2-methyl-5-hydroxy-6: 7-dimethoxy isoflavone which is compared with an authentic sample prepared by an independent method. 5-Hydroxy-7 : 8-dimethoxy isoflavone yields a small quantity of 5-hydroxy 6: 7-dimethoxy isoflavone during alkali fission.
Similar content being viewed by others
References
Krishnamurthy and Seshadri..J.S.I.R., 1953,12B, 120;Proc. Ind. Acad. Sci., 1954,39 A, 144.
Narasimhachari, Row and Seshadri..Proc. Ind. Acad. Sci., 1952,35A, 46.
Rao, Seshadri and Vishwanadham..Ibid., 1949,29A, 72.
Shriner and Stephenson ..J.A.C.S., 1942, 2737.
Ishwar Dasset al...Proc. Ind. Acad. Sci., 1953,37A, 599.
Seshadri and Coworkers..Ibid., 1950,32A, 342; 1951, 33A, 257; 1952. 36A 191.
Schonberg and Badran ..J.A.C.S., 1951, 2960.
Mukerjee and Seshadri..Proc. Ind. Acad. Sci., 1952,35A, 323.
Shady and Soine..J. Amer. Pharm. Assn., 1952,41, 325.
Dhar, Narasimhachari and Seshadri .. Unpublished.
Rajagopalan, Narasimhachari and Seshadri..J.S.I.R., 1952,11B, 347.
Bakeret al. .. Chemistry and Industry, 1953, March 21.
Karmarkaret al. ..Proc. Ind. Acad. Sci.,37A, 660.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mahesh, V.B., Narasimhachari, N. & Seshadri, T.R. Synthetic experiments in the benzopyrone series. Proc. Indian Acad. Sci. 39, 165–175 (1954). https://doi.org/10.1007/BF03048535
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03048535