Abstract
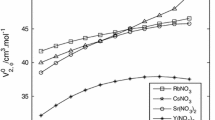
The expansion coefficients of barium, strontium and lead nitrates have been studied above room temperatures. Lead nitrate decomposes rapidly beyond 200° C. while crystals of barium nitrate crack above 270° C. Strontium nitrate exhibited no such phenomena till 550° C.
These crystals are isomorphous. The expansion coefficients of lead and strontium nitrates are nearly equal; they also possess the same ionic radii. Barium nitrate has a considerably smaller expansion coefficient though its ionic radius is larger than for the other two. This peculiar behaviour of barium nitrate indicates a stronger binding in this crystal—a fact which is supported by measurements on the elastic constants, Raman effect, solubilities of these salts and a study of the magneto-optic anomaly of aqueous solutions of these salts.
Similar content being viewed by others
References
Bhagavantam and Krishna Rao..Nature, 1948,162, 740.
—..ibid., 1953,172, 415.
Bhimasenachar and Seshagiri RaoProc. Nat. Inst. Sci., 1950,16, 235.
Couture and Mathieu..Comptes Rendus, 1947,225, 1140.
—..Ann. de. Phys. 1948,3, 521.
Gruneisen ..Handbuch d. Physik., Bd. X, Kap. 1, 1914.
Kopp, H...Phil. Trans., 1865,155, 71.
Mellor..Inorg. & Theo. Chem., 1924,3, 849.
Press, D. C...Proc. Ind. Acad. Sci., 1950,30A, 284.
Ramaseshan, S...Ibid., 1950,31A, 172.
Regnault, H. V...Ann. Chim. Phys., 1841,1 (3), 129.
Vegard, L...Zeit. f. Phys., 1922,9, 395.
Author information
Authors and Affiliations
Additional information
Communicated by Prof. R. S. Krishnan, F.A.SC.
Rights and permissions
About this article
Cite this article
Srinivasan, R. Thermal expansion of nitrates of lead, barium and strontium. Proc. Indian Acad. Sci. 41, 49–54 (1955). https://doi.org/10.1007/BF03047172
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03047172